Conclusions
This analysis of data from the MOBILE study demonstrates that use of CGM results in a rapid improvement in TIR and other CGM metrics reflective of hyperglycemia in adults with T2D on basal insulin without prandial insulin. These improvements were sustained over 3 months and resulted in clinically significant improvements in glycemic outcomes with CGM use compared with BGM use. HbA1c decreased from baseline by 1.2% (13 mmol/mol) at 3 months and this decrease was sustained at 8 months (−1.1% (−12 mmol/mol)) as previously reported in the primary analysis of the MOBILE study,18 and also at 14 months in a subset of participants who continued to use CGM (−1.0% (−11 mmol/mol)).26
It is well established that a reduction in HbA1c of 0.4–0.5% (4.4–5.5 mmol/mol) is clinically meaningful27 28 and strongly correlated with a reduced risk of chronic diabetes-related vascular complications in both people with T1D29 30 and T2D.31 32 Similarly, TIR is also strongly associated with chronic diabetes-related complications.33 Cross-sectional studies, along with secondary analyses of data from the Diabetes Control and Complications Trial (DCCT), have documented this association,33–35 with one analysis by Beck et al35 reporting reduced risk of retinopathy by 22% with every 5% increase in TIR. Thus, the degree of improvement in HbA1c and TIR found in the current analysis and previously published MOBILE study results may suggest benefits for reduced risk on incidence of long-term diabetes-related complications. While the adjusted between-group difference in HbA1c between CGM and BGM decreased non-significantly (data not reported) from 3 months, as presented in this analysis, to 8 months, as reported in the primary analysis of the MOBILE study18 (from −0.6% (−6.6 mmol/mol) to −0.4% (-4.4 mmol/mol)), this occurred due to modest changes (±0.1% (±1.1 mmol/mol)) in HbA1c in both groups between these time points. Therefore, the present 3-month analysis combined with the previously published 8-month18 and 14-month26 results of the MOBILE study suggests that continued CGM use results in rapid and sustained improvements in glycemic management in participants with T2D treated with only basal insulin.
The value of CGM in adults with T2D on insulin was also demonstrated in the Steno2tech study. In this 12-month RCT, 83% of participants used basal insulin without prandial insulin and 17% of participants were on MDI. The study results demonstrated a significant 0.8% (8.7 mmol/mol) and 0.9% (9.8 mmol/mol) greater reduction in HbA1c and a 12.4% and 15.2% greater increase in TIR, at 6 and 12 months, respectively, in those on basal insulin and MDI following use of CGM compared with BGM.19 The Multiple Daily Injections and Continuous Glucose Monitoring in Diabetes (DIAMOND) study, a 6-month RCT in people with T2D on MDI demonstrated a greater reduction in HbA1c by 0.3% (3.3 mmol/mol) with CGM use compared with BGM use.14 Together, the current evidence base highlights the importance of CGM use in managing glycemia in all people with insulin-treated T2D. Expanding CGM availability to those with insulin-treated T2D may be appropriate, considering the clinical benefit. However, it is important to note that there are a number of factors that influence access to CGM beyond clinical benefit, including cost and patient quality of life. Improved glycemic outcomes are associated with fewer emergency department visits and hospitalizations from diabetes-related complications,36 with related reductions in healthcare costs.37 38 The ADA Standards of Care guidelines currently recommend CGM use for all people with insulin-treated T2D and suggest consideration of CGM use for those not treated with insulin.21 Future work may show the benefit of CGM use in non-insulin-treated populations,39 expanding the potential for CGM use along the full spectrum of T2D progression and management.
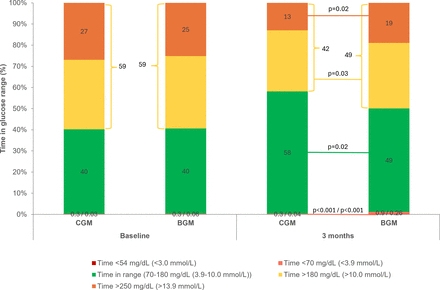
A noteworthy outcome of the present analysis in T2D participants on basal insulin without prandial insulin is the rapid time frame in which clinically relevant improvements in CGM-derived glycemic metrics occurred in the CGM group. In as little as 7 days, significant improvements in TIR, and time >180 mg/dL (>10.0 mmol/L) and >250 mg/dL (>13.9 mmol/L) were observed, which were sustained throughout the 3-month analysis period. These data are consistent with a pooled cohort of T2D individuals on either MDI or basal insulin without prandial insulin, which demonstrated an 11% increase in TIR and decreases in time >180 mg/dL (>10.0 mmol/L) and >250 mg/dL (>13.9 mmol/L) of −10% and −7%, respectively, in 7 days.24 These findings suggest that individuals with T2D at different stages of insulin therapy can experience glycemic benefits very soon after initiating CGM use. The improvement in glycemic outcomes observed in the current study over 3 months is meaningful, since increased TIR is associated with corresponding reductions in HbA1c.11
A strength of this analysis is that it included a population of individuals with T2D receiving diabetes management in a primary care setting who were racially and socioeconomically diverse. As a result, the findings should be generalizable to many individuals with T2D who use basal insulin without prandial insulin. A limitation of the study is that participants had more contact with clinic staff than they would have as part of standard care. This particularly affected the BGM group, which received significantly more interaction than expected in standard care, likely contributing to better outcomes in this study cohort. Additionally, this trial was only conducted within the context of a US-based healthcare system. Nevertheless, other studies exploring real-time CGM use in cohorts from Europe19 and Australia40 demonstrate glycemic benefit for adults with T2D. It is also important to note as a limitation that this post-hoc analysis was not prespecified and exploratory in nature. Due to the lack of difference in changes in daily insulin delivery and diabetes medications between groups, it may be hypothesized that the rapid glycemic improvements in this analysis occurred due to changes in participant behavior. Patient-reported outcomes were not assessed in the first 3 months of the MOBILE study; however, this limited our ability to reveal the mechanisms of rapid glycemic improvements in CGM users and the magnitude of benefits related to changes in diet, physical activity, or medication adherence. Nevertheless, this presents an opportunity for future research in this population.
In conclusion, the results of this 3-month post-hoc analysis of the MOBILE study demonstrate that among adults with T2D with suboptimal glucose levels, CGM use is associated with rapid glycemic improvements in as little as 7 days, with significant improvements compared with BGM without an increase in hypoglycemia at 3 months. These findings add to the growing body of evidence that demonstrates the value of CGM to reduce hyperglycemia in adults with T2D on basal insulin.

Leave a Reply