Discussion
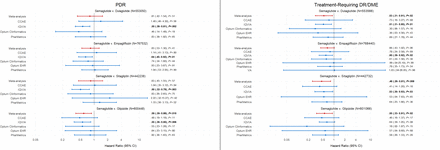
In this retrospective cohort study across 14 databases in the OHDSI Evidence Network, there was no increased risk for either PDR or treatment-requiring DR/DME comparing semaglutide with other GLP-1RAs or non-GLP-1RAs. In the primary active-comparator new-user cohort analysis, there was evidence of a lower risk of PDR when comparing second-line use of semaglutide with glipizide, and a lower risk of treatment-requiring DR/DME when comparing semaglutide with other GLP-1RAs (dulaglutide) and non-GLP1-RAs (sitagliptin, glipizide).
The potential for worsening retinopathy with intensive glycemic control has been known since the 1980s from the landmark Diabetes Control and Complications Trial (DCCT), which studied intensive and conventional treatment among patients with insulin-dependent diabetes. The DCCT assessed retinopathy severity using stereoscopic fundus photographs graded using the Early Treatment Diabetic Retinopathy Study scale. This method of DR assessment is still considered the gold standard in modern clinical trials.47 48 The DCCT found that the risk of significant worsening of retinopathy severity at the 6 and/or 12-month visit was higher among patients assigned to the intensive treatment (13.1%) compared with patients assigned to the conventional group (7.6%).49 50 The magnitude of the hemoglobin A1c reduction was an important risk factor for early DR worsening.49 The Oslo study similarly found that rapid glycemic control was associated with the development of cotton wool spots concerning for transient retinopathy worsening in patients with insulin-dependent diabetes.51 In both studies, these changes appeared transient and were not associated with serious or irreversible vision loss. Importantly, in the long term, the risk of progression of microvascular complications, including retinopathy, was reduced with tighter glycemic and blood pressure control.49 50 52–54 These results are relevant to semaglutide and other GLP-1 RAs, which are considered high-efficacy glycemic therapy, resulting in greater reductions in hemoglobin A1c compared with other second-line T2D medications.55 The potential for early worsening of DR with reductions in hemoglobin A1c as identified in DCCT, combined with findings from SUSTAIN-6 and PIONEER-6 and other studies, has fueled ongoing concern for potential worsening of DR with semaglutide use.3 7 9
Large-scale observational health studies of DR often have to use proxy measures since fundus photographs for grading severity are not typically available. Administrative claims databases do not include retinal imaging, and photos are challenging to share across sites.56 Diagnosis codes to identify DR, without consideration of severity, as has been used in some studies,16–18 is prone to outcome misspecification as less severe DR, for example, non-proliferative DR, is often not coded.57 58 We chose to focus on DR complications—one on a diagnosis of PDR and another on treatment—requiring DR/DME. PDR is typically well coded;57 58 however, by using diagnosis codes alone and not requiring a prior instance of non-proliferative DR, we could be capturing prevalent cases of PDR that were previously treated.59 As a proxy for incident DR complications, we created another definition requiring procedural interventions such as laser, intravitreal anti-VEGF injections and retinal surgeries, as has been done in prior studies.12 The treatment-requiring DR/DME includes not only PDR but also other complications, such as DME requiring intervention. A limitation of both DR outcomes is that we do not know the precise changes in retinopathy staging. The ongoing prospective clinical trial evaluating how semaglutide affects diabetic eye disease compared with placebo among patients with T2D (FOCUS) will help clarify the precise impact of semaglutide on DR staging.60
In the primary analysis, comparing new users of semaglutide as a second-line treatment for T2D with other GLP-1RAs and non-GLP-1RAs, we did not identify an increased risk for PDR or for treatment-requiring DR/DME. There appeared to be a lower risk of PDR comparing semaglutide with glipizide and a lower risk of treatment-requiring DR/DME when comparing semaglutide with other GLP-1RAs (dulaglutide) and non-GLP-1RAs (sitagliptin, glipizide). We chose to restrict our analysis to new users of these medications as second-line treatment after metformin use, based on the American Diabetes Association (ADA) guidelines at the time of study design.55 By restricting our analysis to new second-line users, we are more likely to compare patients with T2D at similar points of their disease course.5 61 Attempting to control for potential confounding from the duration of T2D is critical since it is a major risk factor for both PDR and DME.62–64 By removing the requirement for second-line therapy in one of our sensitivity analyses, we no longer identified a decreased risk of the DR outcomes with semaglutide compared with other GLP-1RAs and non-GLP-1RAs. Not restricting our analysis to second-line users of T2D medications allowed the inclusion of patients with more severe T2D who could be on semaglutide as third or even fourth-line treatment, particularly given the expanding indications of semaglutide for the reduction of cardiovascular and kidney outcomes.1 3 4
The SCCS sensitivity analysis suggests a small increase in treatment-requiring DR/DME associated with semaglutide exposure compared with non-exposure, though elevated rates were also observed with other T2D medications including glipizide. However, these findings should be interpreted cautiously due to potential time-varying confounding by disease duration and severity,43 44 and indications of bias44 in elevated pre-exposure IRR estimates across all T2D medications. The elevated IRR for both DR outcomes in the 30-day window immediately prior to T2D medication exposure could reflect healthcare utilization patterns whereby patients are more likely to be referred for further management of systemic T2D after significant retinopathy is identified. Finally, the SCCS may reflect transient changes in DR paralleling the rapid decrease in hemoglobin A1c associated with semaglutide use3 52; however, these changes are challenging to capture in this analysis due to the inadequate specificity of diagnosis codes for capturing DR progression.57 58 Given these biases, the SCCS findings should be considered exploratory rather than conclusive.
The lack of elevated PDR or treatment-requiring DR/DME risk comparing semaglutide to other GLP-1RAs and non-GLP-1RAs should reassure the prescriber. Our findings are in direct contrast to other observational studies identifying an increased risk of DR outcomes with GLP-1 RA exposure compared with SGLT-2 inhibitors.13 15 These studies were limited to examination of class effects (GLP-1RA) without investigation of drug-specific effects (semaglutide) or only examined patients on GLP-1RA or SGLT-2 inhibitor monotherapy, thus excluding patients prescribed these medications as second-line treatment for T2D as recommended by the ADA.55 Extrapolating findings from DCCT, it should be reassuring that any early worsening is transient in nature and does not affect visual acuity outcomes. With sufficient ophthalmic monitoring, evidence suggests that treating vision-threatening complications (ie, center-involved DME, PDR) after they appear leads to similar vision outcomes as attempting to prevent those complications.48 65 Prescribers of semaglutide and other T2D medications can help ensure that their patients with T2D are closely monitored by eye care providers for the development of vision-threatening complications.
The incidence of PDR and treatment-requiring DR/DME among patients with T2D varies across studies. Using administrative claims from the USA, an estimate for the development of PDR at 5 years following initial diagnosis of T2D is 1.74%, which is similar to our findings.59 A study leveraging administrative claims from Japan estimates the incidence of treatment-requiring DR/DME ranging from 149 to 1722 per 100 000 person-years, depending on baseline characteristics.66 We identified a much lower incidence of PDR and treatment-requiring DR/DME among new users of semaglutide compared with other studies.12 13 The restriction of our analysis on new users of semaglutide as a second-line treatment for T2D and differences in the underlying base populations could be major contributors to a lower incidence rate.
This study has limitations and strengths. Since this was a retrospective large database study, we were unable to assess the precise staging of DR using fundus photographs or other imaging modalities. Although we used a robust and previously leveraged definition of treatment-requiring DR/DME for DR complications, this definition did not capture patients who might require treatment but did not receive it. We were also unable to evaluate whether differences in treatments resulted in differences in visual acuity outcomes. This highlights a broader limitation of many databases for ophthalmic research, as eye examination data are not routinely included, and work is ongoing to standardize these data and incorporate them into the databases to enable future analyses.67 68 Hemoglobin A1c is a major confounder for both PDR and treatment-requiring DR/DME that could not be adjusted for. We also are unable to adjust for duration of diabetes. Despite these limitations, this is, to our knowledge, the largest retrospective observational health study investigating the association of semaglutide with DR complications. Other strengths of the study include its use of sensitivity analyses to test the robustness of our findings against study choices and the use of diverse databases that include both administrative claims and electronic health records.
In conclusion, despite prior evidence and concern for DR worsening, these real-world data do not suggest an increased risk for either PDR or treatment-requiring DR/DME comparing semaglutide with other GLP-1RAs or non-GLP-1RAs. Patients with T2D should undergo close follow-up with eye care providers for DR screening and follow-up, particularly when initiating new medications.

Leave a Reply