Discussion
This multisite cross-sectional study provides novel insights into the complex interplay between hepatic fibrosis, metabolic parameters and ASCVD risk in hospitalized patients with T2DM and MASLD, who had no pre-existing CVD. Our findings suggest that higher SAFE score independently predicted an elevated 10-year ASCVD risk in this high-risk population. Furthermore, we identified hypertension, insulin resistance and LDL cholesterol as significant metabolic moderators of the association between hepatic fibrosis and ASCVD risk. These results advance our understanding of the intricate cardiometabolic interplay in MASLD and carry important implications for ASCVD risk stratification strategies.
MASLD with advanced fibrosis has been strongly associated with a heightened risk of cardiovascular morbidity and mortality, including myocardial infarction, heart failure and all-cause mortality.5 32 Accumulating research suggests that advanced hepatic fibrosis, as quantified by FIB-4, NFS and the AST to platelet ratio index (APRI), represents an independent risk factor for cardiovascular mortality beyond traditional cardiometabolic risk factors in the general population.9 33 34 Notably, Choi et al demonstrated a significant and independent association between these fibrosis indices and CVD mortality.6
However, existing evidence primarily comes from studies on the general populations, including both diabetic and non-diabetic individuals, with a paucity of research focused exclusively on T2DM patients. A previous observational study utilizing Fatty Liver Index and BARD scores to assess hepatic steatosis and advanced fibrosis reported an association between hepatic fibrosis and CVD risk in newly diagnosed T2DM.35 However, this study lacked HbA1c measurements, precluding an evaluation of glycemic control and relied solely on biochemical indices for diagnosing hepatic steatosis, which are less accurate than hepatic ultrasonography. In contrast, our study incorporated comprehensive metabolic parameters, including HbA1c, and used ultrasonography to diagnose hepatic steatosis. Building on recent work by Li et al, which demonstrated the SAFE score’s superior diagnostic accuracy for severe fibrosis and liver-related events compared with FIB-4, NFS and APRI,36 our study revealed that the SAFE score demonstrates a stronger association with ASCVD risk compared with FIB-4 or NFS in hospitalized patients with T2DM. Importantly, this association persisted after comprehensive adjustment for traditional cardiovascular risk factors, underscoring the independent prognostic value of hepatic fibrosis severity as captured by SAFE score. To our knowledge, this is the first large-scale study to examine the association between the SAFE score and ASCVD risk.
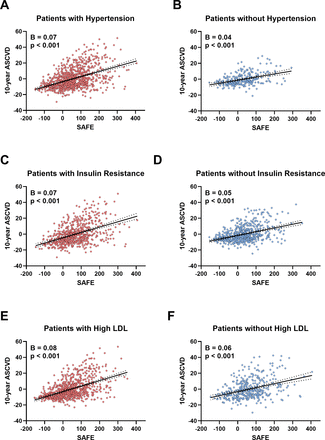
T2DM is intrinsically associated with multiple metabolic abnormalities, including obesity, insulin resistance, dyslipidemia, hypertension and hyperuricemia. Metabolic factors can independently or synergistically contribute to the development of both hepatic fibrosis and CVD.15 To better understand these interactions, we examined the moderating effects of key metabolic parameters on the relationship between hepatic fibrosis and ASCVD risk. We found that this relationship was significantly stronger in patients with hypertension, insulin resistance and with elevated LDL levels, consistent with prior research.37 A meta-analysis identified hypertension as a critical determinant in the link between hepatic steatosis and CVD.38 Furthermore, insulin resistance and hyperlipidemia are known shared pathological mechanisms underlying MASLD and CVD progression.39 40 These mechanisms likely explain our observation of amplified associations between hepatic fibrosis and ASCVD risk in patients with insulin resistance and elevated LDL levels. Collectively, these findings suggest that hypertension, insulin resistance and hyper-LDL-cholesterolemia may function as critical effect modifiers in the hepatic fibrosis–ASCVD risk relationship. Prospective cohort studies are warranted to confirm these observations and unravel the temporal dynamics of these metabolic interactions.
Conversely, traditional lipid parameters (triglycerides and HDL cholesterol) did not significantly moderate the SAFE score–ASCVD risk association. One potential explanation is the high prevalence of statin use among patients, which may have attenuated the variability in triglyceride and HDL cholesterol levels, thereby blunting their moderating effects on ASCVD risk. Additionally, overweight/abdominal obesity was not a significant moderator, likely due to the fact that all participants had T2DM, a key diagnostic criterion for MASLD. The high frequency of obesity in this cohort may have limited the discriminative power in identifying its moderating effect on the hepatic fibrosis-ASCVD link. However, these non-significant findings do not negate the clinical relevance of obesity, dyslipidemia, uric acid, TSH, HbA1c or hs-CRP in CVD risk. Instead, they suggest that the influence of these metabolic factors on hepatic fibrosis–ASCVD interactions may be indirect, masked by stronger metabolic drivers (eg, insulin resistance), or inadequately captured by current categorical definitions.
Clinical implications
The prevention of CVD has become a global health priority, underscoring the need for cost-effective non-invasive evaluation tools. Traditional CVD risk models do not account for hepatic fibrosis, despite its established association with cardiovascular events. Our findings highlight the SAFE score as a potentially valuable metric for ASCVD risk prediction, demonstrating a stronger correlation with ASCVD risk than traditional fibrosis indices. Given its non-invasive nature and superior predictive value, the SAFE score could serve as a clinically actionable tool for identifying high-risk individuals, allowing for earlier cardiovascular intervention strategies in patients with MASLD.
Furthermore, our results suggest that ASCVD risk stratification in MASLD should incorporate key metabolic factors, particularly hypertension, insulin resistance, and LDL cholesterol. A multimodal risk assessment framework, integrating hepatic fibrosis severity with metabolic risk profiling, may enhance MASLD management and optimize preventive strategies against MASLD-associated cardiovascular morbidity.
Pathological mechanisms linking hepatic fibrosis and CVD risk
Hepatic fibrosis and CVD share some pathophysiological mechanisms, such as disruption of lipid metabolic homeostasis, dysregulation of glucose metabolism, accumulation of oxidative stress damage, chronic systemic inflammation, neuroendocrine regulatory abnormalities, prothrombotic state formation and gut microbiota dysbiosis.15 Of particular significance, this hepatic fibrosis–CVD relationship may stem from the progressive deterioration of systemic/hepatic insulin resistance, aberrant secretion of atherogenic lipoproteins and systemic release of multiple proinflammatory cytokines, prothrombogenic factors and vasoactive mediators. Hepatic-derived mediators exacerbate vascular endothelial dysfunction, accelerate atherosclerotic plaque formation, intensify cardiomyocyte metabolic stress and disrupt the coagulation–fibrinolysis balance, ultimately drives the development and progression of CVD.3 41–44 Nevertheless, the precise mechanisms underlying the association between hepatic fibrosis and CVD remain incompletely understood, necessitating further mechanistic investigations.
Study limitations and strengths
This study has several limitations that should be acknowledged. First, the observational design precludes causal inference between SAFE score and ASCVD development. Second, while ultrasonography and non-invasive fibrosis assessment are clinically practical, their diagnostic accuracy remains inferior to liver biopsy histology. Third, our study only included hospitalized patients, potentially introducing selection bias towards individuals with more poorly controlled diabetes. This may limit the generalizability of our findings to community-dwelling populations. Future prospective cohort studies incorporating longitudinal designs and histopathological validation will be crucial in further elucidating the hepatic fibrosis–ASCVD relationship. In addition, although the SAFE score has demonstrated diagnostic utility in general populations, it has not been specifically validated in T2DM populations using liver histology. This limitation should be considered when interpreting our findings. Age was embedded within the formulas for SAFE and ASCVD scores and AST/ALT were embedded within the formula for SAFE. We did not include them as separate covariates to prevent overadjustment and multicollinearity. Thus, findings should be interpreted as the association between these composite measures rather than effects independent of age or liver enzymes.
Despite these limitations, our investigation demonstrates methodological rigor through its multisite design with substantial sample size, comprehensive data integrity and systematic exclusion of patients with active malignancies, hepatic cirrhosis or pre-existing CVD events to mitigate potential confounding effects on outcome interpretation.

Leave a Reply