Study population
This was a retrospective cohort study with patients followed longitudinally. We used electronic health record and associated administrative data from January 1, 2004, through December 31, 2018. The two study cohorts were comprised of patients 65 years or older at the start of enrollment, including VA patients who were dually enrolled in Medicare and KP patients drawn from KP Northwest and KP Mid-Atlantic states.
For eligibility criteria, we defined a diabetes diagnosis as having at least two outpatient or one inpatient diagnosis code (International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM): 250.XX, 357.2, 362.0X, 366.41 and International Classification of Diseases, Tenth Revision, Clinical Modification (ICD-10-CM): E10.X, E11.X, E13.X, where X indicates all subcodes) or a prescription for a diabetes medication. This includes patients with type 1 and type 2 diabetes. Patients were excluded if they had less than four A1c tests or if A1c tests were more than 12 months apart during a 3-year baseline that occurred between January 1, 2005, and December 31, 2017. Patients were removed from the cardiovascular and microvascular outcome analyses, respectively, if they had a prevalent diagnosis for one of these outcomes during baseline (online supplemental figures S1 and S2).
Study variables
Patient-level A1c TIR
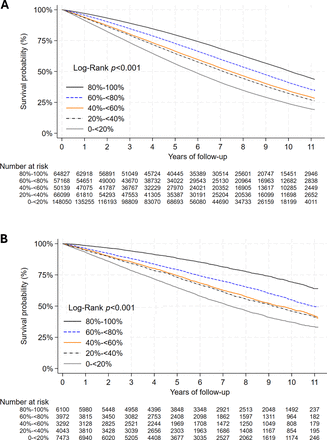
Applying previous methods,16–18 A1c TIR was calculated as the percentage of days during a 3-year baseline that a patient had A1c levels within their individualized target range. To obtain a patient’s A1c target range, we applied the VA/Department of Defense Diabetes Clinical Practice Guidelines12 which proposes four different A1c ranges, 6.0–7.0%, 7.0–8.0%, 7.5–8.5%, and 8.0–9.0%, based on a patient’s life expectancy and the presence and severity of diabetes complications. We estimated life expectancy using a predictive model of clinical and administrative data and separated patients into groups of less than 5 years, 5–10 years, and 10 or more years.19 The Diabetes Complications Severity Index (DCSI)20 was used to identify diabetes complications and their severity. The DCSI is a validated index that scores diabetes complications in seven categories using ICD-9/10 codes and laboratory tests, with a range between 0 and 13. We assessed life expectancy and prevalent diabetes complications for each patient in a 1-year period preceding the baseline to set the initial A1c target range. A1c target ranges and time in range were updated annually during the 3-year baseline to adjust for comorbidities or new diagnoses that impact mortality or severity of complications. A1c TIR was calculated using linear interpolation between A1c values and lab test dates. The total days that A1c values were within the corresponding target range were summed and divided by the total days of baseline (1095 days) and expressed as a percentage. A1c TIR was divided into five categories: 0% to <20%, 20% to <40%, 40% to <60%, 60% to <80%, and 80% to 100% (reference group).
We also studied the direction of out-of-range levels by grouping patients into four mutually exclusive categories based on the percentage of days that A1c was within, below, or above their unique target range. The groups were ≥60% A1c TIR (reference group), ≥60% A1c time below range (TBR), ≥60% A1c time above range (TAR), and a mixed group comprised of patients whose A1c TIR, TBR, and TAR were all below 60%. A≥60% threshold was used because it comprised a majority of time spent in the respective category. For each patient, the sum of A1c TIR, TBR, and TAR equaled 100%.
Clinician A1c TIR as an instrumental variable (IV)
We employed an IV to limit bias from unmeasured factors or confounding in the association between patient-level A1c TIR and outcomes.21 The IV approach helps mimic randomization to allow for less biased estimates. A suitable IV is one that has a strong influence on the likelihood of receiving a treatment but has no direct effect on the outcome and is not associated with confounders.22 Clinical practice variation is commonly used as an IV.23–27 We used clinician A1c TIR as an IV by calculating patient-level A1c TIR at the clinician level. For analysis, patients were aligned to the specific clinician who ordered the most A1c tests for them during the baseline. We then calculated A1c TIR for all the patients assigned to each clinician during baseline and averaged these values. If a clinician had fewer than 10 assigned patients, we used the A1c TIR for all clinicians at the treating site for VA patients and by health system for KP patients.
Covariates
Baseline covariates included age, sex (self-reported), race/ethnicity (self-reported), DCSI Score, comorbidities, medications, laboratory tests, clinician type, calendar quarter in which a patient entered the outcome period, and VA Medical Center or KP health system (online supplemental table S1). The Elixhauser Comorbidity Index28 was used to measure comorbidities during the baseline. Diabetes medications dispensed during baseline were categorized by class. Medication adherence was defined as 80% or greater days with on-hand supply by calculating the medication possession ratio (ie, days’ supply/total number of days) for all prescribed diabetes medications. Laboratory measures included baseline serum creatinine, serum albumin, urine albumin-to-creatinine ratio, and blood lipids (ie, high-density lipoprotein cholesterol [HDL], low-density lipoprotein cholesterol [LDL], triglycerides). Clinical measures included average body mass index and blood pressure during baseline. Multiple measures were averaged and then grouped into categories using clinical criteria (eg, low, normal, high) and a separate category was used for missing values. We also measured the number of A1c tests, the patient’s average A1c level, and A1c SD during baseline. Clinician type included physician, nurse practitioner, physician assistant, or other, and if they were a primary care clinician.
Outcomes
Outcomes were mortality and new cardiovascular and microvascular complications. After excluding patients with prevalent diagnoses during the baseline period, we determined incident cardiovascular outcomes from the DCSI categories for cardiovascular, cerebrovascular, and peripheral vascular diseases and incident microvascular complications from the categories for retinopathy, neuropathy, and nephropathy. To determine all-cause mortality, we used the VA Vital Status File for VA patients, data from the Social Security Administration, state-level vital health status records, and electronic health record information for KP patients.
Statistical analysis
Descriptive analyses were examined for patient-level characteristics and clinical variables. We estimated the effect of A1c TIR on time to death, cardiovascular outcomes, and microvascular outcomes using adjusted Cox proportional hazards regression models. Results from the Cox models were also assessed for violation of the proportional hazards assumption with Schoenfeld residuals. When the proportional hazard assumption did not hold, the effects of A1c TIR on outcomes were estimated with accelerated failure time models. Follow-up began immediately after the 3-year baseline. Patients were censored on death for the cardiovascular and microvascular outcomes, if they disenrolled from the healthcare system, or at the end of the study period. All adjusted models included the main explanatory variables and all covariates. Models were run separately for VA and KP cohorts.
For IV models, we used a two-stage residual inclusion approach. In the first stage, patient-level A1c TIR was regressed on the IV (clinician A1c TIR) and all covariates using a linear regression, applying the criterion that F-statistic less than 10 indicates a weak instrument.29 In the second stage, we included the residual from the first stage as a covariate in a multivariable Cox proportional hazards regression, which was performed separately for mortality, cardiovascular, and microvascular outcomes.
We conducted several additional analyses. A key assumption in IV models is that the instrument influences outcomes only through effects on patient-level A1c TIR. We examined this by studying the association of A1c TIR on mortality, cardiovascular, and microvascular outcomes in patients without diabetes based on the assumption that clinician A1c TIR should not influence their outcomes.30 We created a falsification sample of patients with asthma, chronic obstructive pulmonary disorder (COPD), and heart failure but without diabetes. Using the second stage equations, we predicted outcomes using clinician A1c TIR as an IV and all covariates. Finally, because patients are at risk for diabetes complications and mortality at the same time, we used Fine and Gray’s competing risk model for cardiovascular and microvascular outcomes. Analyses were conducted using STATA V.18.
Data and resource availability
Patient-level data from the Department of Veterans Affairs and KP contain protected health information and cannot be shared. The source code used for generating Elixhauser comorbidities, DCSI index scores, and samples of analytical analyses used for this study will be made available on reasonable request.

Leave a Reply