Methods
This systematic review and meta-analysis was conducted in accordance with the 2020 Preferred Reporting Items for Systematic reviews and Meta-Analyses guidelines and registered in PROSPERO (International Prospective Register of Systematic Reviews) (CRD42023416462).5
Literature search
We conducted a systematic search of PubMed, EMBASE and the Cochrane Central Register of Controlled Trials for relevant papers published from January 2000 to December 2023. The start date of January 2000 was selected as the cut-off, given that RCTs on texting for health promotion were published after this. Medical subject headings and keywords for searches focused on the following three key topics: text messaging, diabetes, and RCTs. Additionally, references from selected articles were manually reviewed to identify relevant citations. The detailed search strategies for each database are provided in online supplemental table 1A–C.
Eligibility criteria
The following inclusion criteria were used to screen for eligible studies: (1) Study type: RCTs; (2) Population: adults aged ≥18 years with type 1 or type 2 diabetes; (3) Intervention: automated text-based or video-based messaging delivered directly to patients via SMS or applications to a phone or tablet device, where the messages promoted glycemic control via awareness of blood glucose trends or encouraging healthy behaviors (eg, physical activity, healthy eating, medication adherence, weight loss); (4) Outcome: mean difference in hemoglobin A1c (HbA1c) between baseline and follow-up in intervention and control groups; (5) Setting: high-income countries as defined by the World Bank (see online supplemental table 2 for a full list of eligible countries)6; (6) Language: study results available in English (text messages could be in any language). To minimize unintentional exclusions, these criteria were manually applied during abstract and full-text screening.
Studies were excluded if messages were only delivered to non-mobile or non-tablet devices (eg, pagers, glucometers, or computer websites requiring login) or if text message generation involved a purely manual process. Compared with automated messages, manual text message generation represents a more labor-intensive and costly intervention with potentially different effects. Studies were also excluded if participants were selected based on a history of cardiovascular events (eg, myocardial infarction, stroke, transient ischemic attack) or if participants had pre-diabetes or gestational diabetes (current or prior) without a diagnosis of diabetes.
Screening and data extraction
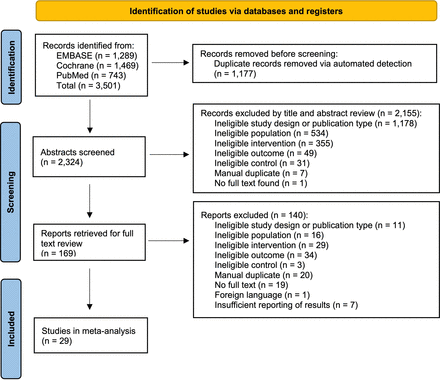
Titles, abstracts, and full texts were screened independently with the Rayyan citation manager by two authors (NP and GSK) in accordance with the predefined eligibility criteria. Data were manually extracted (without automation tools) using full texts to gather study characteristics (first author, year of publication, country, sample size, and type 1 or type 2 diabetes), participant characteristics (age, sex, race and ethnicity, and baseline HbA1c), intervention details (frequency and duration of texting, degree of personalization, presence of bidirectional messaging, and other intervention components), and outcomes (time points and results). Discrepancies in screening eligibility or data collection were resolved via a consensus meeting between NP and GSK. When necessary, consultation was sought by the corresponding author DTK. Where available, within-group means and SDs of HbA1c changes from baseline to follow-up were extracted for intervention and control groups. If a study reported HbA1c results at multiple time points, all were included in the analysis. Results were converted to units of percent HbA1c. For studies reporting only baseline and follow-up HbA1c values, the change in HbA1c between time points was calculated using a correlation coefficient of 0.5, which has been done in similar studies.7
Outcomes
The primary outcome of our study was the between-group difference in HbA1c change from baseline to follow-up.
Risk of bias
Two authors (NP and GSK) independently assessed the quality of included studies using V.2 of the Cochrane risk-of-bias tool for RCTs.8 This tool evaluates potential biases in the following key areas: the randomization process, deviations from the intended interventions (effect of assignment to intervention), missing outcome data, outcome measurement, and selection of reported results. To enhance clarity, we used the online robvis (Risk-Of-Bias VISualization) package to visually summarize risk of bias findings.9
Statistical analysis
Statistical analyses were performed using the meta-analytic functions within Cochrane’s Review Manager (RevMan) V.5.4 and JASP. The between-group difference in HbA1c change from baseline to follow-up was calculated using an inverse variance random effects model. Pooled estimates were expressed as weighted mean differences with 95% CIs. Forest plots were created based on this data. Statistical heterogeneity was assessed using the I2 statistic and Cochran’s Q. Funnel plots and Egger’s regression test were done through the open-source program JASP.10
We conducted two unplanned subgroup analyses. Initially, our a priori protocol intended to stratify studies based on text message frequency. This approach proved unfeasible due to substantial variability in frequency across studies and the absence of a clear cut-off to define a high versus low frequency subgroup. Furthermore, there was an insufficient number of studies per time point to conduct meta-regression with frequency as a continuous covariate. However, we were able to identify alternative subgroups with strong theoretical justification. The first subgroup analysis divided studies based on the study-level mean HbA1c at baseline (≥8.6% vs <8.6%), a cut-off which differentiated participants with poor glycemic control. According to guidelines from the American Diabetes Association and Diabetes Canada, an HbA1c level above 8.5% in elderly individuals is associated with increased risks of long-term harm.11 We hypothesized that text message interventions would have greater benefits for individuals with poorer baseline glycemic control.
The second subgroup analysis compared studies in which texting was the sole intervention component to studies in which texting was combined with other interventions (eg, glucometer, website, telephone calls). This distinction sought to determine whether any observed benefits of texting interventions could be attributed to texting itself.
RevMan was used to conduct a meta-analysis within each subgroup and explore potential sources of heterogeneity.
Overall quality of evidence
The quality of the evidence regarding the effect on glycemic control at 3, 6, and 12 months was assessed using the Grading of Recommendations Assessment, Development, and Evaluation (GRADE) criteria.12 GRADE evaluates several factors that can reduce the quality of evidence including the type of evidence, risk of bias (assessed using the Cochrane risk-of-bias tool),8 consistency across studies, directness of evidence, precision of the estimate, and risk of publication bias. Due to pitfalls in relying on a single method to assess publication bias, both visual inspection of funnel plots and Egger’s regression test were used.13
Sensitivity analysis
We also performed an additional analysis excluding studies at high risk of bias based on the Cochrane RoB 2 tool to evaluate the robustness of our findings.

Leave a Reply