Discussion
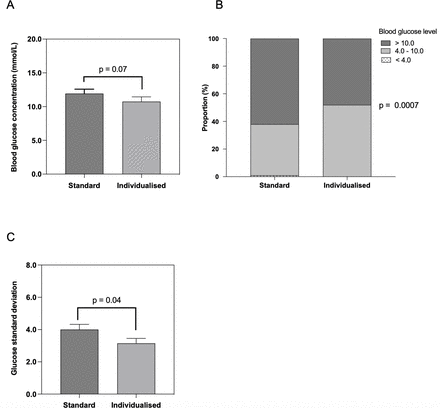
This analysis evaluated the performance of an individualized BBI regimen for the treatment of prednisolone-associated hyperglycemia in hospitalized patients. Compared with patients treated with a standard weight-based BBI regimen, use of an individualized insulin regimen was associated with a mean 24-hour POC glucose on day 1 that was 1.2 mmol/L, but this difference did not reach statistical significance. There was a significantly higher proportion of glucose measurements within the target glucose range and reduced glycemic variability on day 1 of individualized insulin. These results provide proof of concept that individualized BBI treatment of prednisolone-associated hyperglycemia in hospitalized patients may improve glycemic control.
Much of the literature on treatment of prednisolone-associated hyperglycemia focuses on insulin pharmacokinetics; in particular, the concurrent administration of isophane insulin and prednisolone, as the profile of isophane approximates the pattern of prednisolone-induced hyperglycemia.16 25 When extra isophane is coadministered with prednisolone in addition to usual treatment, glycemic control improves.26 27 However, when comparable doses of glargine-based and isophane-based insulin regimens are studied, mean daily glucose levels are similar.17 18 28 29 This suggests that determination of an appropriate insulin dose is more important than insulin pharmacokinetics to optimize glycemic control in patients prescribed prednisolone.
In this study, the mean individualized daily insulin dose prescribed per kg body weight was 23% higher and much more variable than in the standard treatment. Individualized insulin dosing was associated with a lower mean POC glucose that approached statistical significance, but mean blood glucose remained above the recommended blood glucose target range.
In the individualized group, we first calculated a basal insulin dose based on each patient’s overnight insulin requirements during an insulin infusion. This was administered as insulin glargine, as over 80% of patients in the individualized group had type 2 diabetes and commonly need long-acting insulin to optimize glucose control. In our study, there was wide variability in glargine dose (range 0–132 units/day) in the individualized group. As prednisolone predominantly increases glucose concentration during the afternoon and evening, some patients in this group required little or no insulin overnight on the insulin infusion to maintain glucose in the target range. In contrast, in other patients, the overnight insulin requirements were high, likely reflecting the severity of underlying insulin resistance associated with type 2 diabetes.8 20 The average glargine dose of 0.4±0.3 units/kg accounted for 55% of the TDD and was approximately 50% higher than the basal insulin prescribed in the standard group. This resulted in a mean fasting POC glucose of 8 mmol/L with no fasting hypoglycemia, suggesting the amount of glargine prescribed to the individualized group was appropriate.
The total daily prandial insulin dose for the individualized group was also derived from the 24-hour insulin infusion and then split into a 20:40:40 ratio for breakfast, lunch, and dinner to counter the circadian pattern of prednisolone-associated hyperglycemia.16 Across day 1, the average POC glucose concentration in the individualized group remained above the target glucose range at 12:00, 17:00, and 21:00 hours and was not significantly different from the standard group. The lack of benefit is not surprising as our calculation of insulin doses from the insulin requirements on a 24-hour insulin infusion did not result in higher doses of prandial insulin to the individualized group. Our findings are consistent with previous studies using various BBI regimens to treat glucocorticoid-induced hyperglycemia in which postprandial hyperglycemia persisted despite increasing short-acting insulin doses.18 26 27 29 Our findings demonstrate that, despite calculating insulin doses from an insulin infusion, which is a surrogate marker of insulin sensitivity, we underestimated the insulin doses needed to treat postprandial hyperglycemia in patients prescribed prednisolone and highlight the importance of considering higher prandial insulin doses in future studies.
Increased time in the target glucose range is associated with reduced hypoglycemia and 30-day mortality in hospitalized patients.30 Previous studies assessing prednisolone-associated hyperglycemia management reported that different BBI regimens did not significantly affect the proportion of time in the target glucose range, which varied between 19.2% and 66%.17 18 27 29 However, in this study, individualized insulin was associated with a greater proportion of POC glucose levels in the target range. There were no episodes of hypoglycemia in the individualized group, whereas four episodes of hypoglycemia occurred in the standard group. Previous studies have reported both lower18 and higher27 frequency of hypoglycemia with glargine-based BBI regimens. Our study highlights the potential of dosing individualization to reduce the frequency of adverse events such as hypoglycemia and to improve the proportion of glucose measurements within the target range.
Increased glycemic variability and relative hyperglycemia are additional measures of glycemia that are associated with poor outcomes in hospitalized patients.31 In the individualized group, the SD of mean glucose concentration, a measure of glycemic variability, was lower on day 1. Further, the mean SHR in the individualized group was 1.0, which is associated with the lowest rates of mortality in hospitalized patients.31 These positive findings in other glycemic variables further suggest that individualized treatment of prednisolone-associated hyperglycemia may be of benefit.
There were fewer participants in the 2-day analysis, which limits the conclusions that can be drawn. However, there was an increase in glycemic variability across time with no positive change in mean POC glucose or SHR. These results are concordant with previous studies demonstrating that prednisolone-associated hyperglycemia does not generally improve over time, despite increasing insulin and reducing prednisolone doses.17 18 27 29
This study provides proof of concept that an individualized BBI regimen may improve some components of glycemic control in patients with prednisolone-associated hyperglycemia and highlights the importance of adequate prandial insulin dosing in these patients. The individualized group in this study had been prescribed an insulin infusion for 24 hours, and this was used to determine each patient’s daily insulin requirements. We do not necessarily propose that an initial insulin infusion should be standard practice for these patients. Rather, we undertook this analysis to investigate the potential benefit that could arise from individualized insulin dosing for patients with prednisolone-associated hyperglycemia. We have recently reported additional clinical factors to body weight, namely sex, HbA1c, diabetes status, and diabetes treatment prior to hospital admission, that could be used to individualize insulin dose calculations in patients with prednisolone-associated hyperglycemia.19 Taken together, study findings emphasize first the wide variability in basal insulin dose required in this patient group, and the importance of adequate prandial hyperglycemia treatment.
We acknowledge the limitations of the analysis. First and foremost, the use of a historical control group undergoing standard insulin treatment who were older and had a lower eGFR than the individualized group may have impacted glucose levels. Second, there was a difference in how the prandial insulin dose was apportioned and supplemental insulin was only prescribed to the control group. However, the historical control group consisted of patients from the same study site, on the insulin protocol in current clinical use, that is, our hospitals’ current standard of care. Moreover, the difference in prescription of supplemental insulin will result in underestimation and not overestimation of the benefit of individualized insulin. All patients completed the insulin infusion between 09:00 and 16:00 hours, and given the short elimination half-life of Actrapid insulin when administered intravenously, this is unlikely to have affected glucose control the next day, although this is a possibility.32 The subject group comprised patients both with and without known diabetes. While in the future, these patient groups may be managed differently, in hospitalized patients the association between hyperglycemia and mortality is greatest in patients without known diabetes, and current evidence suggests hyperglycemia should be treated regardless of underlying diabetes status.9 31 Additional limitations include a short duration of follow-up and inability to evaluate daily carbohydrate intake; however, the latter is in keeping with standard daily clinical care. Mean prednisolone dose in our study was 30.8 mg/day as this study only enrolled patients prescribed >20 mg/day prednisolone, and this dose is higher than reported in other studies of prednisolone use in hospitalized patients.8 Finally, the sample size was small, particularly for the 2-day analysis. However, the clinical decisions to discharge participants were made by the participant’s treating physician, based on clinical care needs and reflecting routine clinical practice in teaching hospitals.
In conclusion, in this proof-of-concept study, we report that individualizing a subcutaneous BBI regimen for treatment of prednisolone-associated hyperglycemia in hospitalized patients is associated with positive effects on glycemic control. Improved glycemic control was characterized by a modest reduction in mean POC glucose, an increased proportion of blood glucose measurements within the target range, and reduced short-term glycemic variability. In our individualized treatment group, we used insulin glargine as the basal insulin and with a starting dose ranging from 0 to 132 units/day, and this was associated with a fasting glucose within the recommended range for hospitalized patients. The prandial insulin doses prescribed in this study were inadequate to control glucose levels at other times of the day. Given the limitations of the current study, including the use of historical controls and moderate sample size, this data should be viewed as an exploratory, hypothesis-generating analysis. The analysis does support the need for a prospective randomized study comparing the effect of standard weight-based versus individualized insulin dosing on glycemic control in hospitalized patients with prednisolone-associated hyperglycemia.

Leave a Reply