Results
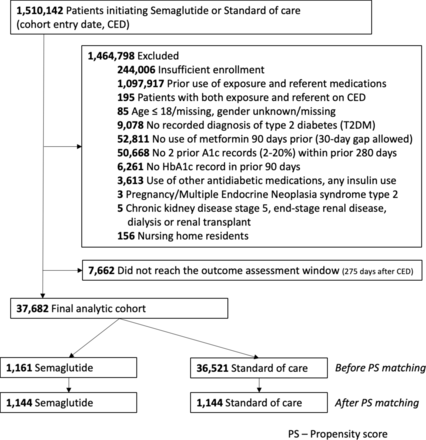
After applying eligibility criteria, we identified 45,344 patients on metformin monotherapy who initiated injectable semaglutide or SoC. Of these, 7,662 (16.9%) did not reach the outcome assessment window for the AS analysis, resulting in a cohort of 37,682 (1161 injectable semaglutide, 36,521 SoC) patients. In the SoC group, there were 19,568 (54.6%) patients prescribed SGLT-2 inhibitors, 6,324 (17.6%) prescribed DPP-4 inhibitors, 6,542 (18.2%) prescribed SUs, and 3,444 (9.6%) prescribed GLP-1RAs (figure 1). Before 1:1 PS matching, semaglutide initiators were younger, more likely to be White, had lower rates of diabetic neuropathy but higher rates of comorbidities such as obesity and mood disorders, and were more likely to be on mood-stabilizing medications. They afforded higher pharmacy copays and were more likely to have commercial insurance. After PS-matching, each group consisted of 1,144 patients, achieving covariate balance with a standardized difference of <0.1 across all variables and a postmatching c-statistic of 0.5 (table 1, online supplemental figure S6). The PS-matched cohort had a mean age of 59 years, with 49% being male and 61% white. Approximately 75% had hypertension, 70% dyslipidemia, and around 40% had obesity recorded (table 1).
Consolidated Standards of Reporting Trials diagram for the as-started analysis of the primary outcome.
Pre-exposure characteristics of study participants before and after propensity score matching for the as-started analysis of the primary outcome
When comparing our matched population to the SEPRA trial cohort, our study population was similar in age, sex, and key comorbidities, such as baseline A1C, dyslipidemia, diabetic neuropathy, myocardial infarction, heart failure, stroke, and thiazide diuretic use. However, our cohort had a higher proportion of non-White patients than the trial and a slightly higher prevalence of diabetic retinopathy, and more use of lipid-lowering medications, beta-blockers, and calcium channel blockers (table 2). Based on the interim results of the SEPRA trial,14 70% of patients completed the study, and the statistical analysis plan indicated that the missing data imputation would be performed for all patients without follow-up A1C measurements, potentially resulting in the imputation of approximately 30% of the values.14 In our emulation, 83% of patients reached the outcome assessment window, with 60% having A1C measurements available, imputing the remaining 40%. The degree of missingness in our study is comparable to the SEPRA trial and aligns with patterns observed in other claims-based database studies, where missing A1C values have ranged from 25% to 70%.10 37–39
Selected patient characteristics from the SEPRA trial and 1:1 propensity score matched database study
Our study found that patients who initiated treatment with injectable semaglutide were more likely to have achieved glycemic control (A1C <7%) (risk ratio (95% CI), 1.30 (1.16 to 1.45)), than those who initiated SoC medications, compared with interim results observed in the trial (risk ratio (95% CI), 1.16 (0.89 to 1.51)). The 1-year reduction in A1C was 0.2% points in this study compared with 0.4% in the SEPRA trial interim results, whereas the number of severe hypoglycemia events resulting in ED or inpatient visits was similarly low in both studies. Based on the trial’s interim results,14 hypoglycemia was reported in <3 participants per arm. We are unable to report exact counts of severe hypoglycemia events in our study due to data use agreements restricting disclosure of cell sizes <11 (table 3). In the trial emulation based on our risk ratio analysis, we achieved concordance on statistical significance and standardized difference but not on estimate agreement. However, in the post-hoc OR analysis, concordance was achieved across all three metrics (table 3). In our secondary—AT analysis, we found that patients treated with injectable semaglutide were more likely to achieve glycemic control than those receiving standard care (risk ratio (95% CI), 1.48 (1.35 to 1.62)). We observed that changes in A1C among patients treated with injectable semaglutide were larger (−1.7% points) than those treated with SoC (−1.2% points) (online supplemental table S4).
Estimated effects for the prespecified primary outcome of the database analysis compared with the SEPRA trial interim results
When comparing injectable semaglutide to individual classes of SoC medications (DPP-4is, SGLT-2is, SUs, and GLP-1RAs), the semaglutide group consistently achieved better glycemic control and showed a slightly greater reduction in A1C (online supplemental tables S5a, b). The largest differences in glycemic control were observed when comparing semaglutide to SGLT-2 inhibitors; however, semaglutide also showed modestly greater effectiveness than other GLP-1 receptor agonists. These findings are consistent with prior systematic reviews, meta-analyses, and network meta-analyses demonstrating superior glycemic efficacy of semaglutide within and across drug classes.40–43
Although computing ORs for frequent endpoints is generally discouraged as it can be a poor approximation of risk ratios,29 30 we conducted a post-hoc analysis using OR, resulting in OR (95% CI) of 1.56 (1.29 to 1.89) in our cohort study, compared with OR (95% CI) of 1.36 (1.03 to 1.78) based on the interim results in the SEPRA trial.
All sensitivity analyses and other post-hoc analyses demonstrated a consistent pattern of results, with patients initiating semaglutide being more likely to achieve glycemic control and exhibiting a slightly higher reduction in A1C. The complete case analysis produced a risk ratio (95% CI) of 1.30 (1.16 to 1.46), which also closely aligned with our primary analysis result (table 3, online supplemental tables S6-S9).

Leave a Reply