Discussion
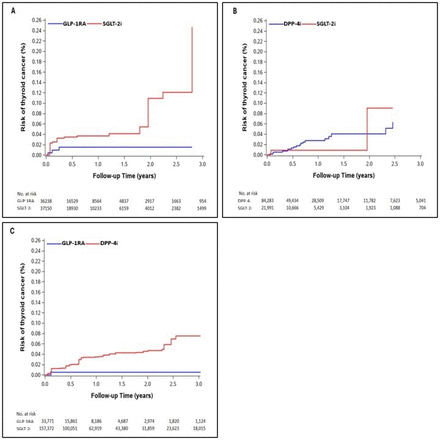
Our study of US Medicare beneficiaries with T2DM suggests that the initiation of incretin-based therapies does not increase the 3-year risk of TC versus SGLT-2 inhibitors, with estimates being null to slightly protective across comparison cohorts. However, the results are too imprecise to exclude a moderate increase in risk. These findings align with prior experimental and non-experimental studies as well as meta-analyses that similarly found no relevant increase in TC risk with the use of incretin-based therapies.9 10 12 13 16–18 This study contributes to the literature as one of the few US-based investigations of TC risk associated with incretin-based therapies. Unlike most prior studies that report HRs, our analysis provides both crude and adjusted cumulative incidence differences (aRDs), offering absolute measures of risk that are directly relevant for clinical decision-making. Our study accounts for death as a competing risk, ensuring valid and reliable risk estimation. By using a large, routinely collected US administrative claims database, our study compensated for the lack of statistical power of the experimental studies to detect rare cancer events such as TC.
Our findings are consistent with recent, well-conducted studies by Pasternak et al and Baxter et al, but offer unique enhancements. Pasternak et al’s Scandinavian study (over 200 000 GLP-1RA users, 4-year mean follow-up) also found no increased TC risk versus SGLT-2i (HR 0.93, 95% CI 0.66 to 1.31).17 Our study adds absolute risk differences (eg, aRD −23 per 10 000), improving clinical interpretability. Similarly, Baxter et al’s multisite study showed no increased risk versus DPP-4i (HR 1.00, 95% CI 0.79 to 1.27) using an intention-to-treat approach.19 We advance this with an as-treated approach to ensure patients were truly exposed, extensive sensitivity analyses, and pairwise comparisons (GLP-1RA/DPP-4i vs SGLT-2i), enhancing robustness and nuance.
Our results, however, contrast several studies—a few observational studies,8 14 15 20 a meta-analysis of experimental studies,11 and preclinical studies in rodents that suggested a potential TC link with GLP-1RA use.4–6 Several possible reasons may explain the inconsistency between our results and the findings from the nonexperimental studies by Bezin et al, Liang et al, Dore et al and Brito et al. Bezin et al conducted a nested case–control study using the French health insurance claims database and reported an increased risk of TC with GLP-1RAs (adjusted HR 1.46, 95% CI 1.23 to 1.74).8 However, this study did not employ an active comparator, which may have introduced confounding by indication. The rationale behind choosing an active comparator is to minimize the impact of confounding by indication and other unmeasured patient characteristics (such as healthy initiator bias or frailty).35 Further, Bezin et al observed an increased risk with a 6-month lag period, which would imply a very short combined induction and latency period for GLP-1RA-induced TCs or may be indicative of a detection bias.36 In contrast, we found no increased risk with a 6-month lag period, and our sensitivity analyses using a 12-month lag period also did not indicate an increased risk of TC, although these results were imprecise. Liang et al performed a retrospective cohort study, supplemented with a nested case–control study using administrative databases from commercial health plans in the US and reported increased risk of TC with exenatide versus other antidiabetic drugs (OADs) (adjusted HR 1.46, 95% CI 0.98 to 2.19).14 Similarly, Dore et al also conducted a cohort study using commercial health insurance claims data and reported increased risk of TC with exenatide versus metformin or glyburide (adjusted RR 1.40, 95% CI 0.80 to 2.40). However, these studies did not employ suitable active comparators. Metformin is often prescribed as first-line therapy in T2DM management, with glyburide as an add-on.37 Thus, comparing exenatide, a GLP-1RA, to metformin or glyburide introduces heterogeneity in disease severity and patient characteristics, which may confound the observed association. OADs, on the other hand, comprise all antihyperglycemics except exenatide with differing mechanisms of action, treatment indications, and side effect profiles. Such heterogeneity in the comparator group complicates the interpretation of results. Notably, these prior studies included younger adults aged at least 18 years (median age<65 years). This difference may hint at potential effect measure modification by age in the relationship between GLP-1RA/DPP-4i and TC. Although Brito et al studied older adults, they did not account for induction and latent periods of cancer diagnosis and development, which might explain the observed increased risk with GLP-1RA use.23 Additionally, TC was defined using only diagnosis codes which likely decreased the specificity of the outcome.
Strengths
Our large observational cohort study used data that are representative of US older adults with T2DM to assess the effect of GLP-1RA/DPP-4i on the incidence of TC. Claims data have many advantages when studying the effects of drugs on cancer outcomes as they include complete longitudinal data on dispensed prescriptions and medical encounters and procedures. Another strength of our study is the use of an ACNU cohort design, which helps to reduce the potential for unmeasured confounding by indication by comparing GLP-1RA/DPP-4i initiators with patients initiating a guideline-recommended clinical alternative. Furthermore, we used rigorous statistical adjustment to minimize remaining imbalances between treatment comparison cohorts. Last, we conducted multiple sensitivity analyses to assess the possibility of residual confounding and obtained results consistent with our primary findings.
Limitations
Our study has limitations that warrant caution in interpreting the findings. The relatively short median duration on treatment (0.82–1.15 years) due to treatment switching or discontinuation during follow-up limits our ability to detect potential TC events associated with long-term use. It does reflect, however, real use patterns, that is, only a few patients with diabetes take incretin drugs over prolonged periods. In settings with short duration on treatment with individual drugs and high frequency of treatment changes as in our study, it is difficult to definitively attribute cancer outcomes to specific treatments without making strong assumptions about induction and latency periods. Notably, results from a sensitivity analysis using the IT approach led to a median follow-up range of 1.42–2.78 years. The IT approach is, however, not ideal for studying safety outcomes as it disregards treatment changes (switching and discontinuation) during follow-up and thus tends to bias results towards the null. Our reliance on claims data to define TC may have introduced outcome misclassification. To address this, we conducted sensitivity analyses using alternative claims-based algorithms that incorporated combinations of diagnostic codes and non-surgical treatment procedures to derive highly sensitive outcome definitions and others that balanced sensitivity and specificity. Results from these analyses were consistent with the primary analysis, requiring thyroidectomy (informed by the American Thyroid Association’s recommendation of surgery as the first step in treatment for all types of TC),38 suggesting that potential misclassification did not meaningfully affect our findings. Differential intensity of medical surveillance may have also introduced detection bias. GLP-1RA users may have had more frequent healthcare encounters, particularly in the context of weight management or cardiometabolic monitoring, increasing the chance of incidental TC detection through imaging or laboratory workup. Although we attempted to assess this by comparing indicators of TC screening intensity across groups, residual differences in diagnostic surveillance may persist. Despite extensive covariate balance achieved via SMR weighting, we cannot exclude residual confounding from unmeasured or poorly measured variables. Claims data lack information on important clinical risk factors like thyroid hormone levels, thyroid nodule characteristics, or family history of thyroid malignancy. These unmeasured factors may have influenced treatment decisions, particularly in light of the FDA black box warning for GLP-1RA regarding a potential risk of medullary thyroid carcinoma. Clinicians may have steered patients perceived to be at higher baseline risk for TC based on, for example, a family history, away from initiating GLP-1RA, leading to a lower underlying risk of TC in the GLP-1RA group, potentially masking an adverse effect. Further, our study reflects real-world patterns of GLP-1RA use among older adults, and the results may thus not be generalizable to younger populations, among whom indications, patterns of use, adherence, and risk profiles may differ.39 Our findings largely reflect use of dulaglutide and liraglutide, which together comprised over two-thirds of GLP-1RA initiations. While caution is warranted in generalizing results to recently approved agents like tirzepatide and semaglutide, the consistency in pharmacologic mechanism supports the plausibility of a class effect. Lastly, the lack of cancer histology information in claims data precluded subgroup analyses by TC histological subtype. This limitation is particularly relevant given that preclinical concerns have primarily focused on medullary thyroid carcinoma (MTC), a rare subtype. Even if histology data had been available, however, the number of incident TC cases in our study was too small to permit analyses by subtype. Any potential signal specific to MTC is likely to be obscured by non-medullary TCs. Thus, our findings cannot rule out a subtype-specific effect, particularly for MTC, which remains an important focus for future research using linked datasets with histopathology data.
Future research should prioritize longer follow-up times and linkage with cancer registries for definitive cancer details and electronic health records to capture additional clinically relevant covariate data. Also, further investigations with larger cohorts to allow precise estimates of effects on medullary TC would enhance understanding of the potential differential impact of incretin-based therapies across cancer subtypes.

Leave a Reply