Discussion
This study provides the necessary analyses and specific information to guide decisions on the required measurement duration when estimating CGM-derived metrics. In contrast to some previous studies,3 6 7 12 the calculations were performed on individual patient data. That individual calculations can lead to different conclusions and decisions for individual patients, for example, within the currently recommended 14-day time window, is underscored by the finding that there was a notable difference between the study population’s mean MAE for TIR estimation versus the population’s maximum MAE, namely, 4.8% vs 13.5%.
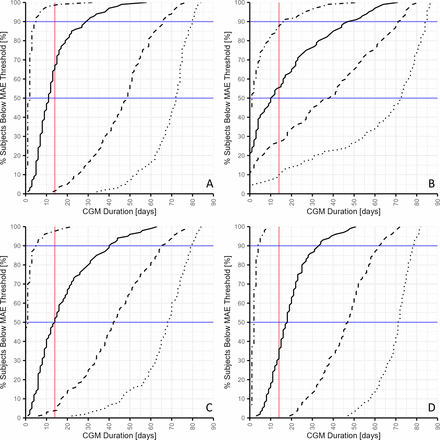
When considering the different arbitrary MAE thresholds, it is relevant to focus on the most commonly applied and evaluated CGM-derived metric, that is, the TIR. It is currently accepted that changes in TIR of at least 5% are considered as clinically significant.5 13 Likewise, a change of 0.5% (5 mmol/mol) for GMI HbA1c can be considered as clinically relevant.14 Then, proceeding with the notion that the MAE of the estimate should not exceed the clinically relevant 5% change in TIR, one could propose the TIR MAE must stay below this 5% threshold. When one assumes that the accuracy of an estimate of long-term glucose regulation must be better than a clinically significant change in TIR for at least 90% of cases, the conclusion from our findings based on the data of all subjects would be that 29 days of measurement are required.
Also, with 29 days of measurement, the CGM duration is sufficient to ensure that 100% of subjects fall below a GMI MAE threshold of 0.5% (5 mmol/mol) and that between 70% and 80% of subjects fall below a TBR MAE threshold of 1%. Although one could argue that every moment of hypoglycemia—and therefore every percentage in TBR—should ideally be detectable, there is no clearly defined clinically relevant change for TBR that the MAE should be below. However, to keep the TBR MAE below 2% for 90% of subjects, a measurement duration of 16 days is already required. If a maximum TBR MAE of 1% is desired, an even longer measurement period is necessary, namely, 51 days. For GV, similar conclusions can be drawn about the influence of CGM duration on the fraction of patients being below MAE thresholds.
When analyzing subgroups, it can be concluded that a CGM duration of more than 14 days is necessary for all but one group for 90% of subjects to be below a TIR MAE of 5%. The exception is the group of hybrid closed-loop system users who needed 9 days to be below the MAE threshold.
All in all, at least in the population currently investigated, we argue that CGM duration longer than the currently recommended 14 days is necessary to ensure an accuracy sufficient for detecting clinically relevant changes in long-term glucose control. However, using the full 90 days is not necessary to secure that more than 90% of the population is within a relevant TIR MAE. When estimating based on shorter CGM durations than the recommended minimum required CGM duration, deviations of the true value may exist and should be considered. On the other hand, when an estimate needs to be made for a shorter time period than 90 days, fewer days will likely be required for a reliable assessment.
Not all findings of this work can be directly compared with existing literature. Herrero et al used MAPE instead of MAE and found a median MAPE of ~19% over 1 day of data.9 Given the fact that the mean TIR for the two datasets used by Herrero et al is ~62%, this would translate to a median MAE of ~11.8%, while a median MAE of 13.4% and a median MAPE of 22.3% were found in our work. For minimum and maximum MAE and longer observation periods, similar deviations can be observed. These small differences could be explained by differences in mean TIR and other characteristics of the datasets.
The strengths of this study are the use of TAs, the number of included patients and the insight into distributions of MAEs within the population. A total of 200 patients with diabetes were included in this study, meaning that the resolution in the fraction of patients below thresholds is 0.5% and therefore individual patients do not have major impact on the outcomes. This work demonstrates that achieving MAE thresholds for every subject requires significantly more time compared with targeting for half of the subjects to be below the thresholds.
Another strength of this study is the use of real-world data. This ensures that sensor usage, therapy, and therefore glucose patterns in the patients resemble those in real-world scenarios. Subjects had various diagnoses, therapies, and sensors. This reasonable reflection of a cross-section of patients with diabetes allows for, if desired, identification of a single minimum required duration of CGM measurement that might be applicable to the entire population. However, as suggested by the results, there are differences between subgroups. Some groups, such as the group with type 2 diabetes and the multiple daily injections group, were relatively small, making the results more sensitive to the influence of individual patients. Moreover, different sensors were used, and sensor placement was not standardized, which may have caused deviations in calculated CGM-derived metrics and might be a limitation of this study. However, sensor placement could also be an unknown factor in scenarios where the results of this study are applied.
A potential limitation of this study is that it does not address the presence of missing data. All CGM data points are concatenated per patient, meaning that a measurement period was reduced to a consecutive data based on the percentage of available data. When applying the results of this study to real-world data, a strategy for addressing this limitation is to concatenate the available data points. For estimation of the minimum required CGM duration, the amount of missing data could be estimated to determine the length of non-concatenated data that is needed to meet the desired MAE. When data are not concatenated and high percentages of data loss or long periods of data loss exist, this could affect the minimum required measurement duration.15 16
The generalizability of the findings needs to be addressed in future research, although both patients with type 1 and type 2 diabetes were represented in the current study, and the population was heterogeneous with respect to age, sensor types, treatment modalities and HbA1c values. Still, the optimal measurement duration may differ, dependent on the characteristics of the population under study. Furthermore, the characteristics of subgroups regarding the relationship between CGM duration and the MAE of CGM-derived metrics should be investigated. It is plausible that comparable MAEs can be achieved with different CGM durations depending on the characteristics of the selected subgroup, for example, the mean TIR, sex or different BMI categories.14 17
Healthcare professionals and researchers using CGM data can take the results of this study into consideration when assessing CGM-derived metrics. When more than 14 days of data are accessible, using them will decrease the MAE of the estimation. The results of this research can be used to give an indication of the fraction of patients being below a MAE for a given CGM duration.

Leave a Reply