Results
Baseline characteristics of the study population (n=10; 4 HNF1A-MODY, 4 HNF4A-MODY, 1 HNF1B-MODY, 1 ABCC8-MODY) are shown in table 1. The study cohort was predominantly non-Hispanic white, with BMIs mostly in the overweight or obese range. Semaglutide was the most frequently prescribed GLP-1 RA, and tirzepatide was the only dual GIP/GLP-1 RA agent prescribed.
•
Demographics, duration, and type of treatment
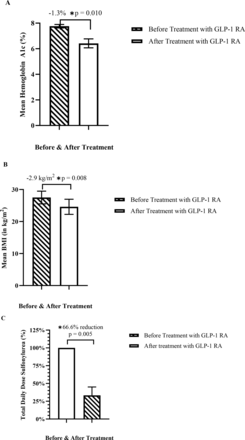
The average age was 39.5±15.1 years, and 80% of patients were female. The mean duration of treatment with GLP-1 RA or dual GIP/GLP-1 RA was 21.4±16.3 months (table 1). Type of GLP-1 RA or dual GIP/GLP-1 RA, duration of treatment, and changes in average Hemoglobin A1c (HbA1c), Body Mass Index (BMI), and sulfonylurea (SU) dose are shown in table 2. In patients with HNF1A-MODY and HNF4A-MODY treated with GLP-1 RA (n=5), there was a statistically significant decrease in HbA1c of 1.3% (95% CI 0.5% to 2.2%, p=0.010), in BMI of 2.9 kg/m2 (95% CI 1.3 to 4.6, p=0.008), and a 66.6% reduction in total daily SU dose (95% CI 33.9% to 99.4%, p=0.005) (figure 1A–C). In patients with HNF1A-MODY and HNF4A-MODY treated with dual GIP/GLP-1 RA (n=3), there was a non-statistically significant decrease in HbA1c of 1.8% (95% CI −0.9% to 4.6%, p=0.104), a statistically significant reduction in BMI of 8.7 kg/m2 (95% CI 4.9 to 12.5, p=0.010). Two patients discontinued SU, and a third patient discontinued insulin (36 units in total) (figure 2A,B). In ABCC8-MODY, GLP-1 RA led to a 1.2% reduction in HbA1c and a BMI reduction of 1.1 kg/m2. In HNF1B-MODY, GLP-1 RA led to a 1.8% reduction in HbA1c and a BMI reduction of 1.2 kg/m2. Notably, both the patients with ABCC8-MODY and HNF1B-MODY did not require treatment with SU or insulin.
Change in HbA1c (A), Change in BMI (B), and Change in Percent of Total Daily Sulfonylurea Dose Before and After Treatment with GLP-1 RA in HNF1A-MODY and HNF4A-MODY. BMI, body mass index; GLP-1 RA, glucagon-like peptide-1 receptor agonist; HNF1A, hepatocyte nuclear factor-1α; HNF4A, hepatocyte nuclear factor-4α, MODY, maturity-onset diabetes of the young.
Change in HbA1c (A), change in BMI (B) before and after treatment with dual GIP/GLP-1 RA in HNF1A-MODY and HNF4A-MODY. BMI, body mass index; GIP, glucose insulinotropic polypeptide; GLP-1 RA, glucagon-like peptide-1 receptor agonist; HNF1A, hepatocyte nuclear factor-1α; HNF4A, hepatocyte nuclear factor-4α; MODY, maturity-onset diabetes of the young.
•
Patients with MODY treatment
All the patients treated with tirzepatide were able to discontinue SU, and reduce or discontinue insulin. There were mild GI adverse effects (nausea (n=1), diarrhea (n=1), and constipation (n=1)) reported in patients who were taking semaglutide 1 mg once weekly and tirzepatide 7.5 mg and 10 mg, respectively, which led to dose reductions (with return to the previous tolerated dose). No severe adverse events, pancreatitis, or hospitalizations were reported.

Leave a Reply