Discussion
In this 26-week, open-label, RCT of HCL versus standard therapy (CSII or MDI without real-time CGM), those allocated to HCL reported significantly greater diabetes-specific positive well-being at 13-week follow-up, which was maintained at 26 weeks. At 26 weeks, the HCL group also reported significantly less frequent perceived unacceptably high glucose levels. Adjusted analyses also showed reduced diabetes distress and fear of hypoglycemia (“maintain high” and “worry” subscales) favoring HCL.
Diabetes-specific positive well-being has not been examined in previous HCL studies and is under-researched generally. The items forming the 4-item scale refer to experiencing a sense of satisfaction from managing diabetes, feeling positive about it, and feeling able to cope with diabetes challenges.16 Gaining this advantage early (within 13 weeks) is an important testament to HCL delivering on the universally high expectations of this advanced technology. This finding also highlights the importance of mid-trial data collection. That it was sustained at 26 weeks, in both unadjusted and adjusted analyses, is also important. Given the relentless nature of diabetes self-management, any improvement in diabetes-specific positive well-being needs to be valued by healthcare professionals, industry, and policymakers. Diabetes-specific positive well-being is worthy of further investigation in future diabetes technology trials.
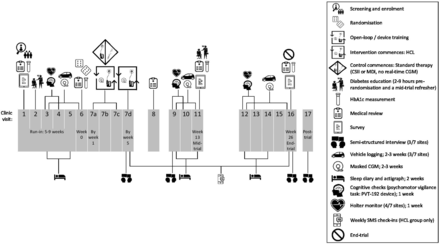
Severe diabetes distress was experienced by about one-quarter of the sample at enrollment, similar to an Australian national survey.22 There was a statistically significant reduction in diabetes distress in the HCL group at 26 weeks, observed only after adjusting for covariates. The fact that the reduction was not significant at 13 weeks, but became evident by 26 weeks in adjusted analyses, likely reflects the complexity of diabetes distress. It may take time or experience with a new technology before distress begins to subside, and there may be interindividual differences in the aspects of diabetes distress that are resolved or exacerbated.23 Qualitative studies highlight that some people using HCL experience hassles, annoyance, and disruption of daily activities (eg, sleep, exercise) due to technical/connectivity problems and alarms.8 9 It is possible that HCL reduced many aspects of diabetes distress, but HCL hassles and trial conditions (figure 1) attenuated such benefits. A 6-month RCT of HCL versus SAP and an 8-month crossover trial of HCL versus SAP both reported no reduction in diabetes distress at follow-up.24 25 However, in our study, the control group continued with their usual management (MDI or CSII without real-time CGM). Thus, the contrast between intervention and control is much greater here, and the between-group difference may be partly attributable to greater visibility of, and improvement in, glucose levels due to HCL. Conversely, three26–28 of four29 12-week single-arm prospective studies demonstrated reduced diabetes distress following HCL or advanced HCL use. Future trials need to examine the effect of HCL on aspects of diabetes distress. For example, in a trial of tubeless HCL, five of the seven subscales of the T1-Diabetes Distress Scale, contributed to lower diabetes distress (powerlessness, diabetes management, hypoglycemia, eating, and physicians), but there was no effect on distress related to negative social perceptions and friends/family.27 We did not analyze the previously identified PAID subscales30 31 because they lack structural validity,30 while the validity of the total score has been replicated consistently.15
Adjusted analyses also show fear of hypoglycemia reduced in the HCL group, in terms of both “worry” (ie, about recognizing or negative consequences of hypoglycemia) and compensatory behaviors to “maintain high” glucose levels. In parallel, we have shown previously that time below range (TBR) was significantly reduced in the HCL group compared with control.6 Qualitative research shows that, when HCL works well, adults with T1D feel peace of mind and less worry about hypoglycemia.8 Therefore, it is possible that the HCL safeguards (eg, warning alarms and insulin delivery modification to reduce severe hypoglycemia risk) likely contributed to reductions in hypoglycemia-related worries and compensatory behaviors to “maintain high” glucose levels. Another 6-month trial showed that HCL reduced such behaviors, but not worries, compared with SAP.24 However, most studies (smaller: N≤58 participants; and shorter: ≤3 months) have reported mixed findings. For example, three single-arm prospective studies observed reductions in fear of hypoglycemia following HCL or advanced HCL,26 28 29 while four crossover trials comparing HCL or advanced HCL to SAP observed no between-group differences.9 25 32 33 The lack of difference may be attributable to the use of CGM in the SAP group. The wider CSII and CGM literature also reports mixed findings regarding the effects of diabetes technologies on the fear of hypoglycemia.11 34
In our study, there were no between-group differences at either follow-up in the HFS-SF “avoidance” subscale, suggesting that HCL did not enable participants to re-engage with activities that may feel risky to them if hypoglycemia occurred. That there was also no improvement in awareness of hypoglycemia symptoms (see below) is of interest, as a recent cluster analysis of the HFS-II has shown that “avoidance” (renamed “restricted activity”) is associated with impaired awareness of hypoglycemia (IAH).35 Our RCT found no significant between-group differences in awareness of hypoglycemia at mid-trial or end-trial. At enrollment, one-third of participants had a score indicative of impaired awareness, which may suggest limited scope for demonstrating population-level improvement. However, we acknowledge this rate is somewhat higher than the average 20–25% reported in most population-based studies.36 Similarly, a crossover trial, also with one-third of participants with impaired awareness, found no differences in awareness between HCL and SAP (across two 4-month periods).25 A 3-month observational study of advanced HCL, including 21% with IAH at baseline, showed significant improvement in hypoglycemia awareness (using the Clarke score) but the reduction in the proportion above the cut-point for IAH (Clarke>3: 21–15%) was not significant.28 Two single-arm prospective studies, conducted primarily with adults, also used the Clarke score, and reported improved hypoglycemia awareness following HCL or advanced HCL.26 29 However, they included a higher proportion of people with impaired awareness at baseline. Further, the Clarke score,37 is known to confound impaired awareness with severe hypoglycemia.38 Thus, score changes may reflect a reduction in severe hypoglycemia rather than an improvement in awareness of symptoms. There is no evidence that technologies (involving some form of CGM) improve awareness of hypoglycemia symptoms, rather, they provide “technological awareness” only while the sensor is worn.39 Thus, restoration of hypoglycemic symptoms may be reliant on psychoeducational approaches, which have shown benefits.40–42
There were no significant between-group differences in total satisfaction with diabetes treatment at either time point in the unadjusted or adjusted models, despite baseline scores not being at the ceiling (ie, scope to indicate increased satisfaction at follow-up). While no previous trials investigating treatment satisfaction have compared HCL to standard therapy among adults, three crossover trials have used DTSQ to examine satisfaction with HCL or advanced HCL versus SAP therapy over 4, 8 or 12 weeks,9 13 32 and two observational studies of advanced HCL have used the DTSQc.43 44 Treatment satisfaction increased only with advanced HCL,32 43 44 suggesting that advanced HCL has improved on earlier iterations of HCL in ways that are meaningful to those using the technologies every day. The DTSQ has also shown greater treatment satisfaction at follow-up in two26 27 out of three single-arm prospective HCL or advanced HCL studies.29 Additionally, we found that HCL participants perceived unacceptably high glucose less frequently at 26 weeks. Relatedly, a prospective study of tubeless HCL (Omnipod 5) has reported less frequency of both unacceptably low and high blood glucose following 3 months of HCL.27
Finally, we also assessed the impact of diabetes on QoL. Our models showed no significant between-group differences at either time point in unadjusted or adjusted models. Three real-world prospective studies, with predominantly adult participants, found improved diabetes-specific QoL following 12 weeks of HCL or advanced HCL, but the lack of control groups are limitations.26 28 29 Conversely, a crossover study among older adults comparing 4 months of HCL and SAP reported no between-group differences in diabetes-specific QoL.25 Above, we have noted the lack of positive impact of HCL on “avoidance” of situations in which hypoglycemia may feel risky, which may be contributing to this lack of benefit for diabetes-specific QoL. In addition, it may be explained by the counterbalancing effect of the potential benefits of HCL against the potential burdens of the uptake of a new diabetes technology combined with the intensive participation requirements of the trial (figure 1).8 9 Importantly, preferences for diabetes technologies are highly personal, thus catering to individual needs and preferences is paramount to optimized and sustainable diabetes self-management.2
Uptake of HCL may improve well-being, treatment satisfaction or diabetes-specific QoL for some people and worsen them for others, depending on their priorities and preferences. Randomization can hide these individual differences. Furthermore, the meaningful effects of HCL on diabetes distress and fear of hypoglycemia after adjusting for known covariates, suggest that the effects were confounded by certain characteristics, and the randomization may not have held for these psychological outcomes.45 For example, fear of hypoglycemia is predicted by prior experience of severe hypoglycemia with traumatic experiences having long-term impacts which may not be fully mitigated by relatively brief access to advanced technologies.41 Therefore, the unadjusted effect estimates for these constructs should be interpreted with caution.
There are noteworthy differences between our former6 and current reports, due to different aims and therefore, different analyses of the data set. First, in the former analyses, treatment satisfaction, and diabetes-specific positive well-being, diabetes-specific QoL, and diabetes distress were secondary outcomes, explored per protocol,14 with adjustment only for score at enrollment. In the current analyses, the models controlled for enrollment and mid-trial scores, and covariates. Second, the current analyses investigated fear of, and impaired awareness of, hypoglycemia, and perceived frequency of unacceptably high and low glucose levels, which were not included in the former report. Third, instead of using log-transformed PAID scores, the current paper presents mean PAID percentage scores. Finally, due to the differing approaches, some results differ. For instance, the current analysis identified improvement in diabetes distress and no change in diabetes-specific QoL at 26 weeks due to HCL. In contrast, the former reported no improvement in diabetes distress and a small improvement in diabetes-specific QoL at 26-week follow-up.
Strengths and limitations
Our RCT represents the largest, longest, and most comprehensive psychological study of HCL compared with standard therapy (without real-time CGM) to date. The strengths and limitations of the overall trial are described elsewhere.6 Many previous quantitative examinations of the psychological impacts of HCL have been limited by small samples, short follow-up, lack of a control group, or lack of adjustment for covariates.3 4 11 Additionally, participants in previous HCL trials are likely to have been “early adopters” of technology, with prior experience with diabetes technologies (eg, CSII or CGM).4 This limits the generalizability of those study findings, as they are unlikely to provide insights into the experiences of people with less access to, or familiarity with, advanced diabetes technologies. Our study has overcome some of these earlier limitations in three ways. First, our study enrolled people whose pre-trial insulin delivery modality was MDI or CSII, (1:1 ratio) and the control group continued with their standard therapy (including no real-time CGM access or allocation to another advanced technology). Second, our large sample allowed for robust analyses controlling for covariates. Finally, our 13 and 26-week follow-ups allowed time for participants to adjust to the HCL and for experiences to be consolidated. Our study compared HCL against standard care (without real-time CGM). At the time of this RCT, only 23% of Australian adults with T1D used CGM.46 Therefore, a non-CGM control group was a justifiable comparator and an appropriate reflection of standard care. While CGM has become the standard of care in the past year, it will continue to be beyond reach for many people with T1D across the world for several years to come.47 Government subsidies can reduce financial inequities to access.48 However, at the time of this RCT, CGM subsidies were not universally available to all Australian adults with T1D. Personal preferences also remain important considerations for the uptake and sustained use of diabetes technologies.49
A potential limitation is that our analyses adjusted for HbA1c, but not time in range (TIR) or TBR, which were excluded from the models due to the data collection timings (figure 1); this would be an interesting area for future research. Psychological data were collected prior to brief diabetes education (provided equally to all participants), while TIR/TBR data were collected after diabetes education. The direct impact of diabetes education was not measured, but at follow-up, glycemic and psychological benefits were observed only in the HCL arm, suggesting technology had a stronger impact than brief education.6 The findings may not be generalizable to all people with diabetes (eg, people attending non-specialist tertiary diabetes clinics). Most participants had a university degree, which is higher than the national average education level,50 higher socioeconomic status,6 and spoke English as their primary language, and the mean HbA1c was near the recommended target (7.8%) at baseline.
The rapid speed at which diabetes technologies are advancing makes conducting and timely reporting of comprehensive evaluations of the latest technology in a RCT setting challenging, as technology is upgraded by the time findings are reported.51 However, our trial provides important and novel insights into the ways in which HCL can affect psychological outcomes. This study focused on a first-generation commercially available HCL device. The translation of these findings remains to be confirmed and further research with other HCL systems (eg, advanced, next-generation, bihormonal, and open-source systems) will increase insights into the fast-growing options available to support T1D self-management.

Leave a Reply