Results
CD8(+) T cells, primarily with a central memory phenotype, are a major component of the lymphoid immune cell population in islets, with no significant differences between type 2 diabetic and non-diabetic donors.
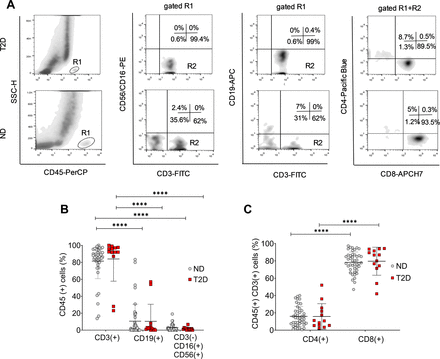
We initially analyzed the lymphoid composition of human pancreatic islets using multicolor flow cytometry on CD45(+) T cells from dispersed islets of 44 non-diabetic and 13 T2D organ donors. The results revealed that CD3(+) T cells were the predominant lymphocyte population in both T2D (84.73±26.45) and non-diabetic islets (81.65±21.40). These CD3(+) T cells were significantly more frequent (p<0.0001) compared with CD19(+) B cells and CD3(−) CD16(+) CD56(+) NK cells in both groups (figure 1A,B). CD19(+) B cells represented a minor fraction of the lymphocyte population, comprising 10.38±20.14 in T2D and 10.58±20.6 in non-diabetic donors. Similarly, CD3(−) CD16(+) CD56(+) NK cells were present in minimal amounts, accounting for 1.61±1.81 in T2D and 2.88±3.75 in non-diabetic donors (figure 1A,B). Importantly, there were no significant differences in the proportions of these cell types between T2D and non-diabetic donors (figure 1B), suggesting that the overall lymphoid composition, with respect to CD3(+) T cells, CD19(+) B cells, and CD3(−)CD16(+)CD56(+) NK cells, remains consistent across diabetic status.
Lymphoid composition in pancreatic islets from ND and T2D organ donors. (A) Representative flow cytometry plots showing the gating strategy used to define the total CD45(+) lymphocyte population by CD45 and side scatter and to identify total CD3(+) T cells, CD3(−)CD16(+)CD56(+) NK cells, and CD19(+) B cells, as well as CD4(+) and CD8(+) T cells in dissociated human pancreatic islets. (B) Cumulative analysis of the proportions of CD3(+) T cells, CD19(+) B cells, and CD3(−)CD16(+)CD56(+) NK from 44 ND and 13 T2D organ donors. (C) Cumulative analysis of the proportions of CD4(+) and CD8(+) T cells within the T cell compartment in dissociated pancreatic islets from 44 ND and 13 T2D organ donors. Data are presented as mean percentages±SD. Each dot represents an individual donor. Differences in the proportion between the groups were compared using the Mann-Whitney test. ****p<0.0001. ND, non-diabetic; NK, natural killer; T2D, type 2 diabetic.
Further analysis of CD45(+) CD3(+) T cells revealed that CD8(+) T cells were the predominant subset in both T2D (79.59±16.16) and non-diabetic donors (78.30±11.94). In contrast, CD4(+) T cells were significantly less prevalent in both groups (T2D: 15.80±14.77, p<0.0001; non-diabetic: 15.96±11.38, p<0.0001) (figure 1C). The comparative analysis of CD8(+) and CD4(+) T cell proportions between T2D and non-diabetic donors showed no significant differences, indicating that the distribution of these T cell subsets is also consistent regardless of diabetic status.
To further assess whether lymphoid composition in pancreatic islets is influenced by obesity, age, or diabetic status, we stratified organ donors by BMI into three distinct categories: normal weight (BMI: 20–24.9; non-diabetic, n=19; T2D, n=6), overweight (BMI: 25–29.9; non-diabetic, n=20; T2D, n=3), and obese (BMI: ≥30; non-diabetic, n=5; T2D, n=4). We analyzed the proportions of CD3(+) T cells, CD19(+) B cells, CD3(−)CD16(+)CD56(+) NK cells, CD4(+) T cells, and CD8(+) T cells across these groups. Notably, our analysis revealed no statistically significant differences in these populations, indicating that lymphoid composition in pancreatic islets is not significantly influenced by BMI or diabetic status (online supplemental figure 1A–E and online supplemental table S4). To further evaluate the potential impact of age, we stratified donors into two age intervals: 45–60 years and 61–81 years. Consistent with our findings from the BMI-stratified analysis, no significant differences in lymphoid cell composition were observed between T2D and non-diabetic donors within these age intervals (data not shown). Collectively, these findings demonstrate that the lymphoid landscape in pancreatic islets remains unaffected by BMI, age, or diabetic status.
To characterize the differentiation stages and memory status of CD45(+) CD3(+) T cell populations within pancreatic islets, we performed a detailed analysis of CD45RO and CD27 surface expression in dispersed islets from T2D donors and non-diabetic controls. Cells were classified into four distinct subsets: CD45RO(−) CD27(+) naive (TN), CD45RO(+)CD27(+) central memory (TCM), CD45RO(+)CD27(−) effector memory (TEM), and CD45RO(−)CD27(−) terminal effector (TTE). We quantified their proportions among total CD4(+) and CD8(+) T cells. Specifically, islets from 34 non-diabetic and 13 T2D donors were analyzed for the CD4(+) T cell memory compartment, while islets from 38 non-diabetic and 13 T2D donors were evaluated for the CD8(+) memory compartment. Our analysis revealed that the majority of CD4(+) and CD8(+) T cells exhibited a TCM and TEM phenotype in both T2D and non-diabetic donors, with TCM populations being predominant. In contrast, the frequencies of naive and terminal effector cells were significantly lower (figure 2A–D). There were no significant differences in the frequencies of TCM, TN, TEM, and TTE populations between T2D and non-diabetic donors (online supplemental tables S5 and S6).
Differentiation stages and memory status of CD45(+)CD3(+) T cell populations in pancreatic islets from ND and T2D organ donors. Representative flow cytometry plots showing naive and memory subpopulations of (A) CD4(+) T cells and (B) CD8(+) T cells. (C) Cumulative analysis of the proportions of CD45RO(+)CD27(+) TCM, CD45RO(−)CD27(+) TN, CD45RO(+)CD27(−) TEM, and CD45RO(−)CD27(−) TTE phenotypes within the CD4(+) populations in dissociated islets from 34 ND and 13 T2D donors. (D) Cumulative analysis of the proportions of CD45RO(+)CD27(+) TCM, CD45RO(−)CD27(+) TN, CD45RO(+)CD27(−) TEM, and CD45RO(−)CD27(−) TTE phenotypes within the CD8(+) populations in dissociated islets from 38 ND and 13 T2D donors. Results are presented as box and whisker plots showing the median. Each dot represents an individual donor. Differences in the proportion between the groups were compared using the Mann-Whitney test. *p<0.05, **p<0.01, ***p<0.001, ****p<0.0001. ND, non-diabetic; T2D, type 2 diabetic; TCM, central memory T cells; TEM, effector memory T cells; TN, naive T cells; TTE, terminal effector T cells.
CD8(+) T cells in T2D donors have phenotypic characteristics of resident cells
To further characterize CD8(+) T cells in dispersed islets, we analyzed tissue-specific resident memory cell markers CD69 and CD103 in samples from five non-diabetic and five T2D donors. Our findings revealed that all T2D donors exhibited significantly higher expression of CD69(+)CD103(+) markers (69.4±7.37) compared with CD69(+)CD103(−) (20.4±13.4, p<0.0001), CD69(−)CD103(+) (5.08±1.78, p<0.0001), and CD69(−)CD103(−) (3.76±2.02, p<0.0001). This indicates that most CD8(+) T cells in the pancreatic islets of T2D donors display a TRM phenotype. Similar proportions of non-circulating CD8(+) T cells expressing the canonical residency markers CD69(+)CD103(+) (68.6±8.08, p=0.87) were also observed in the islets of non-diabetic donors (figure 3A,B).
Characterization of resident CD8(+) T cells in pancreatic islets from ND and T2D organ donors. (A) Representative flow cytometry plots showing the expression of CD69 and CD103 on CD8(+) T cells in islet cell suspensions. (B) Cumulative analysis of CD8(+) T cells expressing the CD69 and CD103 from five ND and five T2D organ donors. Data are presented as mean percentages±SD. Each dot represents an individual donor. Differences in the proportion between the groups were compared using the Mann-Whitney test. ****p<0.0001. ND, non-diabetic; T2D, type 2 diabetic.
T2D donors exhibit a higher proportion of glucagon-producing alpha cells but maintain similar proportions of insulin-producing beta cells compared with non-diabetic donors
Quantitative assessment of the composition of endocrine cells within the pancreatic islets showed that the percentage of glucagon-producing alpha cells was significantly higher in T2D donors (23.11%±14.98%, n=13) compared with non-diabetic donors (15.49%±9.51%, n=44, p=0.03). Conversely, the proportion of insulin-producing beta cells did not differ significantly between T2D donors (18.92%±10.68%, n=13) and non-diabetic donors (23.61%±12.99%, n=44, p=0.24) (figure 4A,B).
Percentages of pancreatic insulin and glucagon-positive cell populations in pancreatic islets from ND and T2D organ donors. (A) Representative flow cytometry analysis of islets cell suspension depicting the percentage of insulin and glucagon-positive cells. (B) Cumulative analysis of glucagon-producing alpha cells and insulin-producing beta cells from 44 ND and 13 T2D organ donors. Data are presented as mean percentages±SD. Each dot represents an individual donor. Differences in the proportion between the groups were compared using the Mann-Whitney test. *p<0.05. ND, non-diabetic; T2D, type 2 diabetic.
Decreased glucose-stimulated insulin secretion in type 2 diabetic islets
To evaluate islet functionality, we assessed insulin secretion in response to glucose stimulation in islets from 44 non-diabetic and 13 T2D organ donors. At a basal glucose concentration of 1 mM, T2D islets exhibited insulin release levels comparable to non-diabetic islets, indicating that basal insulin secretion is not significantly impaired in T2D islets. However, upon stimulation with a high glucose concentration of 16.7 mM, T2D islets released significantly less insulin than non-diabetic (p=0.02) (figure 5), indicating that glucose-stimulated insulin secretion is significantly impaired in T2D islets.
Insulin secretion in pancreatic islets from 44 ND and 13 T2D organ donors after stimulation with 1 mM glucose (basal secretion) and 16.7 mM glucose (stimulated secretion). Data are presented as mean±SD. Each dot represents an individual donor. Differences in the proportion between the groups were compared using the Mann-Whitney test. *p<0.05. ND, non-diabetic; T2D, type 2 diabetic.

Leave a Reply