Methods
Study population
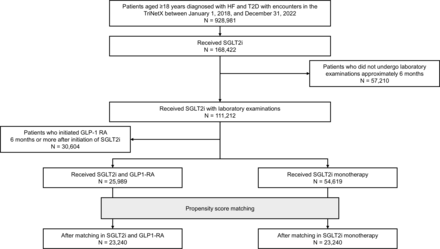
We conducted a multicenter, retrospective observational study using TriNetX (TriNetX, Cambridge, Massachusetts, USA), a global healthcare data and analytics platform. The TriNetX platform provides real-world data analysis using electronic health records (EHR) for more than 250 million patients from 130 healthcare organizations across North America, South America, Europe, the Middle East, Africa, and the Asia-Pacific region. The TriNetX database aggregates de-identified longitudinal health data from millions of patients across multiple healthcare settings, including hospitals, outpatient centers, and primary care clinics. Each healthcare organization maintains local control of its data, whereas TriNetX securely accesses and aggregates de-identified data across institutions. By only aggregating the results of the analysis, TriNetX enhances privacy. Available data included patient demographics (such as age, sex, race, and ethnicity), diagnoses (standardized by the International Statistical Classification of Diseases and related Health Problems (ICD)-10 codes), procedures (using Current Procedural Terminology codes), laboratory results, prescribed medications, and clinical outcomes. Because the TriNetX platform collects data from the EHR, disease diagnosis is not based on diagnostic criteria but rather on the recording of disease names in the EHR; therefore, we identified disease and health problems from the ICD-10 code. We included patients aged ≥18 years with HF (ICD-10 code: I50) and T2D (ICD-10: E11) between January 1, 2018, and December 31, 2022. The exposure group was defined as patients who initiated combination therapy with an SGLT2i and a GLP-1 RA. Combination therapy was defined as the initiation of a GLP-1 RA within 6 months after starting SGLT2i therapy. The control group was administered an SGLT2i monotherapy. Patients who received a first-time prescription for a GLP-1 receptor agonist or an SGLT2 inhibitor were included. To minimize immortal time bias, patients who underwent laboratory examinations approximately 6 months after initiating SGLT2i therapy were included according to this additional criterion. The date of the laboratory examination was defined as the index date for the observation period.
Outcome measures
The primary outcome measure was all-cause death during the 1-year follow-up period. The secondary outcome measure was hospitalization. As one of the limitations for the TriNetX platform, it is not allowed to determine cause‐specific death or hospitalizations.
Data collection and definitions
Covariates considered confounding factors for exposure and outcome were extracted from the database with reference to previous studies.20 21 Each factor was extracted from 1 month before day 0 of the index date. Comorbidities were defined as conditions with a recorded diagnosis within 1-month prior to the index date. Diseases recorded in the EHR during the observation period were included as comorbidities, whereas previously recorded diseases that were discontinued at the beginning of the observation period were not included as comorbidities. In addition, conditions documented more than 1 month before the index date were also considered comorbidities if they remained active during the 1-month period following the index date. Diagnoses that had been previously recorded but were deleted more than 1 month before the index date were not considered comorbidities. We extracted the following 43 factors: age, sex, body mass index (BMI), race (Asian, American Indian or Alaska Native, black or African American, Native Hawaiian or other Pacific Islander, white, other race, unknown), diagnosis (hypertensive heart disease, ischemic heart diseases, old myocardial infarction, cardiomyopathy, atrial fibrillation and flutter, peripheral artery disease, hypertension, hyperlipidemia, chronic kidney disease, cerebral infarction), medication (beta blockers, ACE inhibitors, angiotensin II receptor blockers, sacubitril, mineralocorticoid receptor antagonists, diuretics, loop diuretics, thiazides, calcium channel blockers, platelet aggregation inhibitors, anticoagulants, biguanides, sulfonylureas, dipeptidyl peptidase 4 (DPP-4) inhibitors, insulins, statins), laboratory assay data (sodium, potassium, glomerular filtration rate (GFR), hemoglobin, albumin, low-density lipoprotein (LDL) cholesterol, hemoglobin A1c (HbA1c), B-type natriuretic peptide (BNP), N-terminal pro-B type natriuretic peptide (NT-proBNP), and systolic blood pressure, diastolic blood pressure, heart rate, left ventricular ejection fraction (LVEF). A list of definitions of the covariates is provided in online supplemental table 1.
Ethics approval
This study was conducted in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology Reporting Guidelines. This study used TriNetX and did not require ethical review, primarily because it deals only with anonymized data; the results available for reference are representative of the study population and do not deal with individual patient-level data.22 23 This study was approved by the Ethics Committee of the Omi Medical Center (local identifier: 2024–0044) because it included patient data from our institution, although the data were completely anonymized, as mentioned above.
Statistical analysis
Covariates were compared between patients who received SGLT2i and GLP-1 RA and those who received SGLT2i monotherapy. Categorical variables are presented as numbers (%), and continuous variables are presented as means with SD. To reduce the potential confounding effects in the comparison between patients who received SGLT2i and GLP-1 RA and those who received SGLT2i monotherapy, we estimated a propensity score (PS) using a logistic regression model that adjusted for all covariates. One-to-one pair matching between patients who received SGLT2i and GLP-1 RA and those who received SGLT2i monotherapy was performed using greedy nearest neighbor matching without replacement, using calipers of width equal to 0.1 SD of the logit of the PS. To measure the covariate balance, we checked the standardized mean difference (SMD) of before and after matching. When the SMD was <0.10,24 25 there was negligible imbalance between the two groups. The cumulative incidence of the outcome measures that occurred during the 1-year follow-up period was estimated using the Kaplan-Meier method, with intergroup differences assessed using the log-rank test. Patients are censored on the day after the last recorded clinical event within the analysis time window. Person-time at risk was calculated from the index date until the earliest occurrence of the outcome, loss to follow-up, or the end of the study period. Incidence rates were expressed as events per 100 person-years, calculated by dividing the number of events by the total person-years of follow-up. These analyses were conducted for both all-cause death and hospitalization. The risks for patients who received SGLT2i and GLP-1 RA relative to those who received SGLT2i monotherapy for the outcome measures were estimated by calculating crude HRs and their 95% CIs. To assess the potential impact of unmeasured confounding, we calculated E values based on HRs and their corresponding 95% CIs for outcome measures.26 27
To measure the differences in treatment effects, subgroup analyses were performed for HF (HFrEF (ICD-10 code: I50.2 or I50.4), HFpEF (ICD-10 code: I50.3 or I50.4)), age (18–64 years, 65–74 years, over 75 years), sex (female, male), BMI (<30 kg/m2, 30 kg/m2 ≤), HbA1c (<7.0%, 7.0–7.9%, 8.0% ≤), GFR (<60 mL/min/1.73 m2, 60 mL/min/1.73 m2 ≤), ischemic heart disease, and hypertension. The TriNetX platform required the creation of additional cohorts for subgroup analysis. After PS matching, the difference in treatment effects between patients who received SGLT2i and GLP-1 RA and those who received SGLT2i monotherapy on all-cause death was evaluated within each subgroup. Crude HRs with the corresponding 95% CIs were calculated. Similarly, a subgroup analysis was conducted for hospitalization.
Cohort definitions and statistical analyses were performed on May 14, 2025, using the Query Builder and Analytics Functions on the TriNetX platform. All tests were two-tailed, and differences were considered statistically significant at p<0.05.

Leave a Reply