Discussion
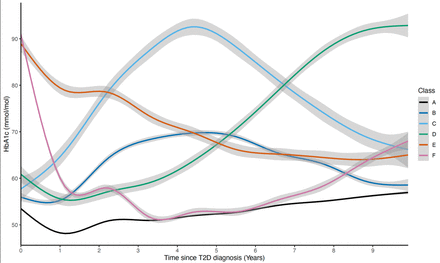
In this study, we used the extensive data available in the UKB and identified six classes of individuals of European ancestry with T2D based on their HbA1c trajectories over a mean follow-up of 8.1 years from diagnosis. The reference class (A; “low and stable”) represents a trajectory previously observed in other studies. This class has the lowest initial HbA1c levels of all classes and experiences a dip around 1 year after diagnosis, before rebounding and slowly increasing thereafter.13 15 Classes B (“low parabolic”) and C (“high parabolic”) represent individuals who experience increases in HbA1c during the first 5 years post-diagnosis, followed by a decline, with Class C experiencing a more pronounced increase and subsequent decline than Class B. Classes E (“slow decrease”) and F (“rapid decrease”) contain individuals with high HbA1c levels at diagnosis, that decrease over time, with Class F having a higher initial HbA1c and a steeper decline. Class D (“steep increase”) has a moderately high initial HbA1c, with a rapid increase approximately 4 years postdiagnosis. Overall, 23.2% of participants were members of Classes B–F (non-reference/atypical classes), characterized by higher and more variable HbA1c trajectories compared with the low and stable trend observed in Class A (reference/typical class).
Younger age of T2D onset and higher fasting blood glucose at T2D diagnosis were associated with class membership for nearly all classes, compared with Class A. While the association with fasting glucose is expected due to its strong correlation with HbA1c, the link between younger age of T2D onset and class membership indicates that individuals who develop T2D at a younger age tend to have poorer glycemic control and a higher risk of developing complications. Possible explanations for this include having higher adiposity, showing lower adherence to T2D medications, and/or having a genetically distinct phenotype which is associated with different risks of T2D complications.29 30 This finding aligns with previous studies, which have identified younger age of T2D onset as a risk factor for developing vascular complications.31
In the all-classes combined analysis, higher BMI polygenic score was associated with non-reference class membership, but phenotypically measured BMI was not. This could suggest a pleiotropic genetic effect of the BMI polygenic score on body fat composition, which is strongly associated with the development of T2D and complications.32 33 Additionally, the BMI polygenic score may be capturing changes in BMI that occur alongside the HbA1c trajectories which may not be captured by a single phenotypic BMI measurement.34
In the healthcare utilization analysis, we found a strong link between belonging to a non-reference class and reduced GP visits before T2D diagnosis. Since increased GP visitation is associated with lower hospitalization rates in T2D patients and early diagnosis is crucial for glycemia management, these results suggest that lower healthcare utilization prior to diagnosis could lead to a more advanced T2D at diagnosis, poorer glycemic control, and higher risk of postdiagnosis complications.35
The time-to-event analysis of secondary outcomes showed that all non-standard classes were at increased risk of developing all disease outcomes, except MDD, compared with Class A. While the association between atypical HbA1c trajectories and risk of T2D complications is well established, we identified three classes (C, D, E) at notably elevated risk of diabetes-related complications and all-cause mortality. The observed differences between Classes E and F (slow vs rapid decrease) demonstrate total exposure to high HbA1c levels and early glycemic management as important risk factors for the development of complications, as reported previously.36 37 Similarly, the increased HbA1c variability when comparing Classes B and C (low vs high parabolic) underscores the need for longer-term and more intensive HbA1c management on the observation of rapidly increasing HbA1c. Class D may reflect poor response to second-line T2D therapies or therapeutic inertia, where delayed treatment adjustments lead to worse long-term glycemic control and higher complication risk.38
This UKB study of T2D HbA1c trajectories offers several advantages over previous research. First, the strict T2D definition increases the likelihood of including only true T2D cases. Second, the large sample sizes, stringent model selection, and flexible time modeling ensure that LCGMM-generated trajectories represent meaningful HbA1c subclasses.39 Third, we consider a broad range of exposures and secondary outcomes, allowing a comprehensive investigation of how these trajectories develop and affect the clinical course of T2D in the UKB. Finally, this is the first study to incorporate genetic information into LCGMM, revealing a novel association between the BMI polygenic score, but not BMI, and atypical HbA1c trajectories, alongside a class-specific association between the T2D polygenic score and Class F.
However, our study has several limitations. First, LCGMM is computationally intensive. It is therefore possible that with more computational power, a model with more classes could have been selected. However, this is unlikely as the improvements in goodness of fit and class stability by including more classes were small after inclusion of a sixth class (online supplemental table 3). Second, there is a healthy volunteer bias in the UKB, meaning these individuals may not be truly representative of the UK-based T2D population. A higher proportion of T2D patients may therefore be assigned to non-reference classes in a more representative UK sample. Cohorts that do not rely on individual-level recruitment, such as the Clinical Practice Research Datalink, should be used to validate our results. Third, T2D diagnosis criteria have changed substantially over the past two decades, with increased population-wide T2D screening and monitoring. This could introduce cohort-specific effects related to the start date of medical records in the UKB. Fourth, despite the large number of participants in our study, only 8% and 4% had prevalent and incident MDD respectively, limiting our ability to fully determine the relationship between HbA1c class trajectories and MDD. Finally, due to limited sample size for individuals of non-European ancestry, and the lack of availability for well-powered genetic studies in individuals in this group, we excluded individuals who were non-European. As there is known heterogeneity by ancestry for T2D disease progression and outcomes,40 further studies should focus on extending these methods to individuals of non-European ancestry.
In conclusion, six classes with distinct HbA1c trajectories were identified in 12,435 individuals of European ancestry with T2D, using the UKB linked to primary care data. Participants in non-reference classes (23%) had increased risk of developing T2D complications, including all-cause mortality. Younger age at T2D diagnosis and lower healthcare utilization prior to diagnosis were identified as important risk factors for following an atypical trajectory. This suggests that the risk of developing complications is related to lower healthcare utilization prior to T2D diagnosis. Additionally, although all individuals in this study that followed an atypical trajectory were at higher risk of developing complications, we identified ∼7% of participants (Classes C, D, E) who displayed highly accelerated T2D progression, as defined by a greater degree of medication transition and a higher risk of developing T2D complications and all-cause mortality. In summary, this study shows that participants who diverge from the typical T2D HbA1c trajectory can be identified as targets for intensive intervention.

Leave a Reply