Discussion
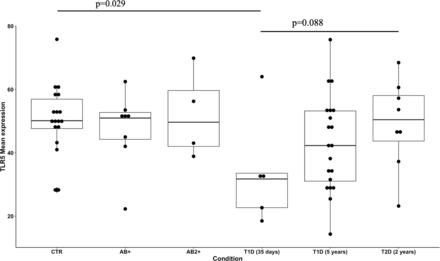
In the present study, we have demonstrated a significant difference in TLR5 expression in the islets of Langerhans between patients with newly onset T1D and healthy controls, and the TLR5 expression correlated inversely to islet-infiltrating CD3 T cells in the same patients, as well as in diabetic NOD mice. Additionally, we identified a TLR5-related polymorphism associated with T1D. TLR5 expression was partially normalized in patients with T1D with a duration of 5 years. At that time, the destructive diabetogenic inflammation has largely completed, and the patients are most often in a stable condition when having an appropriate insulin treatment.
Gram-negative bacteria with flagellin stimulate TLR5, which enhances granulocyte activity to eliminate these bacteria. Experience with flagellin due to low hygiene standards seems natural in Russian Karelia, which also has low incidence of T1D.18 The hygiene theory is indeed all about exposure to activated bacteria, which, via TLR5-related mechanisms, may explain the lower incidence of autoimmunity.19
Bacterial infections are not an obvious job for T cells which are present to react against tumor cells and virus, and to aid the production of antibodies via T helper and B lymphocytes. Importantly, the development of T1D does require islet destruction by T cells; this is supported by many studies, for example, treatment of patients with T1D with CD3 antibodies is beneficial as it reacts against aggressive T cells.20
Internal factors such as pregnancy can influence TLR5 gene expression, leading to its upregulation.21 This may take place in order to prevent T cells from destroying the fetus which expresses paternal antigens. Interestingly, the beta cells produce 40% more insulin during pregnancy, reflecting a high activity. We have previously demonstrated that such high activity is associated with an increased risk of developing T1D.4 22 Yet, despite the seemingly increased risk, there is no increase in the incidence of T1D during the first two trimesters.22 In the third trimester, there is a 3.8 times increased risk of developing T1D but not more, despite the even higher pressure on the beta cells during this time.22 These observations align well with our results, highlighting the potential implications the TLR5-regulated T cell activity may have on T1D.
A highly interesting point is whether the TLR5 expression can be influenced by exogenic factors. It has been described that bacteria can invade the ductus pancreaticus and be found in the pancreas of humans.23 These include Staphylococcus aureus,23 which has been reported to inhibit the expression of TLR5.24 Fusidic acid, an antibiotic targeting Staphylococcus, has also been shown to reduce the incidence of diabetes in BioBreeding (BB) rats.25 Additionally, Helicobacter pylori warrants mention, as its presence has been shown to increase T1D incidence in humans by 1.77 times26 and is able to inhibit TLR5 by masking its expression.27 H. pylori is often present in the stomach and duodenum, but no studies have examined whether it is present in the pancreas. Thus, highly relevant exogenous factors related to T1D risk exist, which can affect TLR5 expression.
Polymorphisms of the TLR5 gene have been associated with innate immune responses in other inflammatory diseases as well, including systemic lupus erythematosus.28 Notably, the SNP rs5744168 of TLR5 generates a stop codon that could affect TLR5 function,29 whereas rs113292043 is an intergenic variant with no known functional consequences. In addition to T1D, we have also shown that the same polymorphisms correlate with reduced number of monocytes, increased number of neutrophils, and rs113292043 with reduced T1D risk.
It should be acknowledged that the DiViD material consisting of living patients is very small (n=5). This restricts the generalizability of the findings, and confirmation in larger, multicenter cohorts is needed. The connection between TLR5 and T cells may not be finally settled, but the results in this study strengthen the association. It is also supportive that an effect of low TLR5 has been found to enhance development of diabetes in NOD mice.9 Furthermore, a higher diabetes incidence has been described in germ-free male NOD mice that have had no opportunity for experiencing TLR5 stimulation.30
In conclusion, a treatment strategy against early development of T1D could involve flagellin to increase TLR5 expression, and thereby naturally inhibiting T cells and potentially avoiding autoimmunity.

Leave a Reply